The Auditory System
The General Organization of the Auditory System An Introduction to the Physics of Sound
Sound travels in waves through the atmosphere and are measured in decibels (dB), a logarithmic scale of measurement of sound pressure level. Approximate scale values are as follows: a whisper is about 30 dB, a loud conversation about 50 dB, a jet taking off about 120 dB, a howitzer (artillery firing) is about 150 dB. There are physical dimensions of sound (frequency and wavelength) and psychological dimentions (pitch; a function of experience, culture and musical training) and timber (complex interaction of fundamental and overtone series that modulate one another harmonically to produce unique combinations of overtones). The behavior of sound waves in the air is complex and involves pitch, timbral and echoic cues. Rarefaction and condensation waves use sound emitted by an object to estimate departure and approach. Doppler shifting is the physical correlate of these departing and approaching wavefronts.
The Auditory System Mediates the sense of hearing. The sensory experience described as hearing is as broad as the sound spectrum itself. From signals of impending danger, like a car horn, to the pleasing sounds that fill a concert hall, much of our daily behavior is determined by the sounds around us. The auditory system is also our principal communication portal, allowing us to understand speech. This system, like the somatic sensory and visual systems has a topographic organization determined by the peripheral receptive sheet. And similar to the other systems, the auditory system consists of multiple parallel pathways that engage multiple cortical regions either directly or via complex corticocortical networks. Each auditory pathway is hierarchically organized and has the connections and properties to mediate different aspects of hearing.
The complexity of the auditory pathways derives from the particular properties of natural sounds, with their diverse frequency characteristics, multiple sources of origin, and large dynamic ranges. However, an added measure of complexity is imposed on the human auditory system by the demands of understanding speech. Although the physical characteristics of a spoken word may not be any more complex than many nonlinguistic sounds, the linguistic quality of the stimulus engages unique cortical areas.
Anatomy of the Ear
The ear consists of the outer ear (pinna), middle ear (external auditory meatus) and inner ear (cochlea) where the transduction (processing) of sound occurs. and where the sensory receptors for hearing are located. Sound waves are captured by the outer ear which are funneled into the external auditory meatus which functions as an impedance transformer that facilitates transmission of airborne sound into vibrations of the fluid in the cochlea. The middle ear is filled with air and acts as a cushion behind the tympanic (drum) membrane. Its proper function depends on air pressure in the middle ear cavity being equal to the ambient pressure, facilitated by the opening and closing of the eustachian tube which connects from each ear bilaterally to the posterior roof of mouth. The incus, malleus, and the stapes are the smallest bones in the body and transcribe sound to the cochlear instrument. The stapes performs like a piston with in and out motion that sets the fluid of the cochlea in motion. The skull also conducts sound (bone-conduction) which is useful for low frequencies however, neural transmission representation in the cochlea pushes beyond the physical limits of bone conduction.
Transduction of the auditory pathway
Sound as a stimulus (rarefaction and compression of air and resulting waveforms) activates the tympanic membrane. Mechanical transduction (middle ear ossicles) matches tympanic and hydraulic impedance. Hydraulic transduction (middle ear to endolymph) converts mechanical signals to fluid displacement. Genesis of generator potential (hydraulic to electrical transmission) involves a fluid displacement creating a micromechanical wavefront. The action potential (conversion of generator potential to action potential) is caused when mechanical shearing deforms hair cell cilia. Complex behavior of hair cells reflects mechanical, electrical, neural and muscle-like properties. Relation of basilar membrane structure and hair cell tuning. Hair cell ciliary linkages are spring-like structures signaling length and displacement of hair cells. Electrical tuning of hair cells. Na+ channels in hair cells are localized in specific hair cell membrane subregions.
The process of hearing begins on the body surface, as sounds are conducted by the auricle and external auditory meatus to the tympanic membrane. Mechanical displacement of the tympanic membrane, produced by changes in sound pressure waves, is transmitted to the inner ear by tiny bones termed the middle ear ossicles. The inner ear transduction machinery is located within the temporal bone in a coiled structure called the cochlea. This is the location of the auditory receptors, termed hair cells because they each have a cilia or hair bundle on their apical surface. Each auditory receptor is sensitive to a limited frequency range of sounds. Hair cells in the human cochlea are not bitotically replaced, and their numbers decline throughout life. This reduction results from conditions such as ear infections, exposure to loud sounds or drugs with ototoxic properties, and aging.
The cochlea is remarkably compact and its central projections are strongly divergent. Spiral ganglion cells sit in small clusters embedded in the temporal bone. Some physiological properties of auditory nerve axons; specific afferent frequency tuning, distinct response profiles distinguish VIIIth (vestibulocochlear) nerve fibers, primary-like discharge pattern, build-up units increase the number of spikes/time period, onset units fir most at stimulus onset, other types also exist and this diversity has important theoretical implications for thinking about how hair cells behave and for the possible existence of ganglion cell subtypes.
The Auditory Nerve
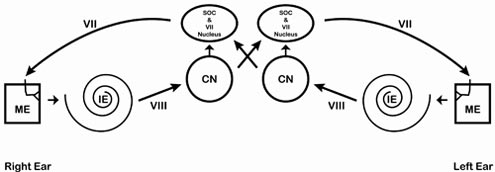
The Vestibulocochlear nerve (VIII) is a sensory nerve for hearing and balance and is the only purely sensory nerve subserving two modalities or two separate components. The Vestibular Nuclei receive input from vestibular hair cells via vestibular ganglion cells for equilibrium analysis and project to the forebrain, the oculomotor system and the spinal cord. The cochlear nuclei receive input from spiral ganglion cells that innervate cochlear hair cells. The vestibular component innervates the semicircular canals, saccule, and utricle and mediates balance. The cochlear component innervates the organ of Corti and serves hearing. The Vestibulocochlear VIII nerve leaves the medulla at the cerebellopontine angle to enter the inner ear as two parts, the cochlear division for hearing and the vestibular division for maintenance of equilibrium.
1. The very great majority of fibers present in the auditory nerve innervate inner hair cells.
2. Single fibers of the auditory nerve are always excited by auditory stimuli and never show sustained inhibition to single stimuli.
3. The fibers have lower thresholds to tones of some frequencies than of others. The relation between threshold and stimulus frequency is known as the "tuning curve". Tuning curves show one threshold minimum, at what is known as the characteristic frequency. The threshold rises sharply for frequencies above and below the characteristic frequency. The tuning curve therefore shows a sharp dip in this frequency region. Tuning curves of auditory nerve fibers are similar to the tuning curves of hair cells and of the mechanical response of the basilar membrane.
4. The great majority of (90%) of auditory nerve fibers have minimum thresholds in a 10dB range near the animal's absolute threshold. The others have thresholds spread over a 60dB range above that. The low-threshold fibers have particularly high rates of spontaneous activity in the absence of sound.
5. Fibers show a sigmoidal (S-shaped curve) relation between firing rate and stimulus intensity. Low-threshold fibers also have steeper rate-intensity functions, often going from threshold to maximum rate in 20-30dB (the dynamic range) at any one frequency. Fibers of higher threshold have shallower rate-intensity functions and wider dynamic ranges, up to 60dB or more.
6. The frequency-resolving power of auditory nerve fibers has been measured by a "quality" factor, by analogy with a quality factor for filters. The quality factor is the characteristic frequency divided by the bandwidth of the fiber to tones at an intensity 10dB above the best threshold. This is called "Q10" or "Q10dB". Therefore, fibers with a high Q10 have good frequency selectivity. At any one frequency, different fibers have Q10s in a restricted range. In any one animal, the range of Q10s at one frequency is twofold or less. In cats, the greatest Q10s are reached at around 10kHz, where they have an average value of eight. The Q10s match, to a first approximation at least, the Q10 of the basilar membrane vibration.
7. During tonal stimulation, auditory nerve fibers fire preferentially during one part of the cycle of the stimulating waveform if the frequency of the stimulus is below 4-5kHz. The fibers are excited by deflection of the basilar membrane only in one direction.
8. For fibers with characteristic frequencies below 4-5kHz, clicks preferentially evoke responses at certain intervals after the stimulus. A histogram of action potentials made with respect to time after the stimulus suggests that the fibers are activated by the half cycles of a decaying oscillation of the mechanical resonance on the basilar membrane. The frequency of the oscillation is equal to the characteristic frequency of the fiber.
9. One tone can reduce, or suppress, the response to another, even though single tones are only excitatory. This is called two-tone suppression. The suppression arises from the non-linear properties of the basilar membrane mechanics. Two-tone suppression can also be seen in the basilar membrane mechanics and the responses of inner hair cells. Stimuli other than tones cause suppression too.
10. One stimulus can mask the response to another. Masking mainly occurs because the masking stimulus produces a greater firing rate than the masked stimulus. The other mechanisms of masking are the suppression of the response to one stimulus by another, and adaptation in the fiber produced by an ongoing stimulus.
11. When two-tone stimuli are used, auditory nerve fibers can respond to distortion products as a result of non-linear interactions in the cochlea. One distortion tone, known as the cubic distortion tone, is at a frequency 2f1-f2, where f1 is the lower of the tones presented and f2 the higher.
Auditory nerve fibers do not provide a simple representation of the basilar membrane input/output function. Auditory nerve fibers can show either steeper or shallower rate-level functions after sensorineural hearing loss. Thus compression algorithms that are designed to overcome a reduced dynamic range may need to account for more than reduced basilar membrane compression. this is particularly true in cases of mixed inner hair cell and outer har cell damage where ihc and ohc damage may interact to produce different effects on an responses.
noise induced hearing loss can produce very steep auditory nerve growth at high levels
anatomy of a neuron; dendrites, cell body (soma) and an axon
afferent hair cell; 90-95% of AN synapse w/ inner hair cells each is innervated by multiple; "many to one". 5% outer hair cells synapse afferently termed one to many; one to ten
Types of Fibers: Type 1 - large, myelinated, 95%, inner hair cells; bi-polar cells
Type 2 - small, myelinated and unmyelinated, innervate the ohc; uni-mono
tonotopicity of the auditory nerve: arrangment is a bullseye, with low in the middle and high on the outside
response/characteristic frequency: responsive to single tones and always elicit an excitatory response, never inhibitory
each fiber has a low threshold at one frequency, the characteristic frequency
Tuning Curves: When you record the post-stimulus time histogram, you get a tuning curve
place code & phase locking: if phase-locking is integrated over a population, the code could be reconstructed
The Cochlea
A topographic relationship exists between the location of a hair cell in the cochlea and the sound frequency to which the receptor is most sensitive. As discussed below, from the base of the cochlea to the apex, the frequency to which a hair cell is maximally sensitive changes systematically from hight frequencies to low frequencies. This differential frequency sensitivity of hair cells along the length of the cochlea is the basis of the tonotopic organization of auditory reception. Many of the components of the auditory system are tonotopically organized. The topographic relationship between the relationship between the receptor sheet and the central nervous system is similar to that of the somatic sensory and visual systems, where the subcortical nuclei and cortical areas have a somatotopic or retinotopic organization. In each of these cases, the topographic organization of the central representations is determined by the spatial organzation of the peripheral receptive sheet. An important difference exists however. The receptor sheets of the somatic sensory and visual systems are spatial maps representing stimulus location. The cochlea represents the frequency of sounds. Sound source localization is counted by central nervous system auditory neurons located in the spiral ganglion. The central processes of the bipolar neurons from the cochlear division of the vestibulocochlear nerve (cranial nerve VIII). These axons project to the ipsilateral cochlear nuclei, which are located in the rostral medulla.
The cochlea is a hollow, fluid-filled tube (Reissner's membrane) within which the basilar membrane resides
The air-borne vibrations displace perilymph, which in turn transmits the waveforms to the endolymph and thus moves the hair cells apical tips against the overlying basilar membrane, creating a receptor potential
Structure of the basilar membrane is directly related to it's mechano-electrical performance
The hair cells
1. The cochlea is a coiled tube, divided lengthways into three scalae. The three divisions are known as the scala vestibuli, the scala media and the scala tympani. The two outer scalae, the scala vestibuli and the scala tympani contains perilymph, which is like normal extracellular fluid in composition, and is at or near ground potential. The scala media contains endolymph, which is more like intracellular fluid, and has a positive potential (+80mV). The positive potential is generated as a diffusion potential across the intermediate and basal cell layers of the stria vascularis, driven by concentration gradients arising from Na+/K+-ATPase and Na+/2Cl-/K+-ATPase ion pumps in the stria vascularis.
2. The organ of Corti contains the auditory transducers. The organ sits on the basilar membrane dividing the scala medai from the scala tympani. The transducing cells are called hair cells. Hair cells are of two types, known as inner and outer hair cells. They have many hairs, or stereocilia, projecting from their apical (apex) surface. Deflection of the hairs initiates transduction, via a pull on the tip links between the stereocilia, which opens the mechanotransducer channels near the tips of the stereocilia by a direct mechanical action. The molecular identity of the mechanotransducer channels is not known.
3. Deflection of the hairs is produced by deflection of the basilar membrane. The latter occurs as a result of a sound-induced displacement of the cochlear fluids, which interacts with the stiffness of the basilar membrane, to produce a progressive traveling wave on the membrane. The wave passes up the cochlea from base to apex.
4. Traveling waves produced by sounds of high frequency come to a peak near the base of the cochlea. High-frequency sounds are therefore transduced near the base of the cochlea. The traveling wave produced by low-frequency sounds comes to a peak further up the cochlea, and low-frequency sounds are transduced near the apex.
5. The traveling wave has a sharply tuned peak. The sharp peak underlies the sensitivity and frequency selectivity shown by the rest of the auditory system, and indeed the sensitivity and frequency selectivity shown by the whole organism. The sharply tuned peak arises because the outer hair cells, when stimulated by the movement, make an active mechanical response which amplifies the vibration of the basilar membrane as the traveling wave passes through. The traveling wave therefore increases in amplitude as it passes along the cochlear duct, until it dies away abruptly when it reaches a point where the cochlear partition can no longer sustain vibrations of that particular frequency. This point is nearer to the base for high-frequency stimuli and nearer to the apex for low-frequency stimuli.
6. The active amplification has its largest effect at low stimulus intensities. At higher intensities, it makes a smaller contribution. The result is that the basilar membrane moves with a compressive non-linearity, that is the response does not grow as fast as the input. This has an important functional consequence, because it allows the auditory system to discriminate stimuli over a very wide range of stimulus intensities.
7. Deflection of the stereocilia by the traveling wave, by opening and closing ion channels in the stereocilia, modulates the current being driven into the hair cells by the combined effects of the positive endocochlear potential (+80mV) and the negative intracellular potential. The current produces potential changes that can be measured in the hair cells with fine microelectrodes, and grossly in the cochlea with larger electrodes.
8. Inner hair cells have resting potentials of about -45mV. They produce both an a.c. voltage and a steady d.c. depolarization in response to sound. The potentials have sharp tuning curves and amplitude functions similar to those shown by the basilar membrane vibrations. Outer hair cells have resting potentials of about -70mV. They show a.c. potential changes in response to sound and, depending on the circumstances, either no, or a depolarizing, or a hyperpolarizing, d.c. response. Like inner hair cells, they show sharp tuning curves and amplitude functions similar to those shown by basilar membrane vibrations.
9. The potential changes in inner hair cells serve to govern the release of neurotransmitter glutamate, to produce action potentials in the auditory nerve fibers. Outer hair cells amplify the mechanical traveling wave, producing a mechanical motile response in the hair cells. The motile response may arise from one or both of the following: (1) a lengthwise contraction and expansion of the outer hair cell body in response to intracellular depolarization and hyperpolarization or (2) Ca2+ ions entering through the open transducer channels, which may change the conformation of the mechano-transducer apparatus, leading to an output of mechanical energy that can feed back into the mechanical system.
10. The cochlear microphonic is the extracellular correlate of the a.c. current flowing through the hair cells. It is generated predominantly by outer hair cells. The summating potential is primarily the extracellular correlate of the d.c. component of the current flowing through hair cells. Depending on circumstances, it can be recorded as either a positive or negative shift in the scala media and probably receives contributions from both inner and outer hair cells, and possibly from other sources.
11. The massed synchronized activity of auditory nerve fibers, called the compound action potential (CAP) or the N1 and N2 potentials, can be recorded with gross electrodes in response to stimulus onsets.
perilymph and endolymph: perilymph is mostly Na+ and at or near ground potential 0V
endolymph is mostly K+ ions and is at or near +80mV
this creates the endolymphatic potential
stria vascularis produces endolymph
Organ of Corti: approximately 35mm
inner hair cells form a single longitudinal row running along the inner side of the sensory region with respect to the center of the spiral - short and fat
Hair Cell Transduction: when the stereocilia bundle is deflected, the tip link stretches and pulls open the mechanotransducer channel
The Traveling Wave: The response of the basilar membrane underlies the performance of the whole auditory system
Determinants of Resonant Frequency:
The Cochlear Amplifier: Prestin!
ultimately modulating the mechanotransduced action potential
THE COCHLEA IS NOT PASSIVE, IT PRODUCES ENERGY!
boosts up the response to up to 60dB!
Sharp Tuning:
EVOKED POTENTIALS:
reproduces frequency and waveform of a sinusoid perfectly
generated from the outer hair cells
Auditory System Cochlear Nerve
The cochlear nerve (part of the VIII cranial nerve) conducts impulses generated in the organ of Corti to the cochlear nuclei; the central pathways from these nuclei travel to intermediate cell stations of the brain stem and thalamus and on to the cerebral cortex.
The central processes of the spiral ganglion neurons form the cochlear nerve, which leaves the petrous portion of the temporal bone via the internal auditory meatus and runs a short course to the cerebello-pontine angle. On entering the rostral medulla, each axon of the nerve bifurcates, sending a branch to each of the cochlear nuclei, dorsal and ventral. These nuclei are found in the rostral medulla and the caudal pons. The dorsal and ventral nuclei are situated posterolateral and anterolateral to the inferior cerebellar peduncle, respectively.
Efferents from the dorsal cochlear nucleus form the dorsal acoustic stria to cross the pontine tegmentum to a contralateral relay cell complex, the superior olivary nucleus. Giving a collateral to the olivary nucleus, the efferent fibers contribute to the ascending lateral lemniscus. The lateral lemniscus is the major brain stem auditory pathway; it is located in the lateral tegmentum, from the rostral medulla to its termination in the inferior colliculus and the medial geniculate body. Some ascending dorsal cochlear efferents in the lateral lemniscus terminate in the nuclei of the lateral lemniscus, arranged adjacent to and along the extent of the lateral lemniscus; others continue on to the ipsilateral inferior colliculus on that side.
Efferents from the ventral cochlear nucleus may project to the ipsilateral dorsal cochlear nucleus or arc around the inferior cerebellar peduncle. The latter form the intermediate acoustic stria to the ipsilateral or contralateral superior olivary nucleus. These fibers continue on as part of the lateral lemniscus to terminate in the lateral lemniscal nuclei or the ipsilateral inferior colliculus.
Efferents from the ventral cochlear nucleus may also form the ventral acoustic stria or trapezoid body, running transversely through the caudal pons. Collaterals of these efferents terminate in the ipsilateral superior olivary nucleus or in the nuclei of the trapezoid body. The trapezoid nuclei and the neighboring ventral cochlear efferents and their collaterals constitute the trapezoid body complex . The trapezoid body fibers may synapse in the contralateral superior olivary nucleus or bend rostrally to contribute to the lateral lemniscus and ascend to the lateral lemniscal nuclei or inferior colliculus. No fibers from the cochlear nuclei ascend in the ipsilateral lemniscus.
In the midbrain, most of the lateral lemniscal fibers terminate in the inferior colliculus, an important relay center for impulses particularly concerned with auditory reflexes, such as involuntarily jumping in response to a loud noise. From each inferior colliculus, some axons pass to the contralateral colliculus via the commissure of the inferior colliculus; other efferents form the brachium of the inferior colliculus and project to the medial geniculate body of the thalamus.
Neurons of the medial geniculate body send axons to the superior temporal gyrus of the cerebral cortex. These axons form a large bundle called the auditory radiation, composing the sublenticular part of the internal capsule and the lateral part of the corona radiata. The axons of the auditory radiation terminate in area 41 (M; Heschl's gyrus or the primary auditory cortex) on the superior temporal gyrus.
In addition to this extensive afferent pathway, there is also an efferent tract, the olivocochlear bundle. This tract projects to the hair cells of the organ of Corti from contralateral brain stem nuclei, including one of the nuclei of the superior olivary complex. Such a pathway represents an inhibitory feedback mechanism on auditory input; stated in more familiar jargon, we can "turn off" what we don't want to hear.
Destruction of the cochlear nuclei or nerve causes complete deafness on the affected side. Lesions of the lateral lemniscus or area 41 cause minimal bilateral deafness, a condition difficult to detect clinically because of the extensive crossed and uncrossed fibers shown here.
The Subcortical Nuclei
The cochlear nuclei consist of the ventral cochlear nucleus, which has anterior and posterior subdivisions, and the dorsal cochlear nuclus. Neurons in these three components have distinct connections with the rest of the auditory system and give rise to parallel auditory pathways that serve different aspects of hearing. A key function of the anteroventral division of the cochlear nucleus is horizontal localization of sound The posteroventral division contributes to a system of connections that regulate hair cell sensitivity. The two divisions of the ventral cochlear nucleus project to the superior olivary complex, a cluster of nuclei in the caudal pons. Most neurons in the superior olivary complex project via an ascending pathway called the lateral meniscus to the inferior colliculus located in the midbrain. The projection from the ventral cochlear nucleus to the inferior colliculus is bilateral. The dorsal cochlear nucleus is thought to play a role in identifying sound source elevation. It projects directly to the contralateral inferior colliculus, also via the lateral lemniscus. The inferior colliculus is the site of convergence of all lower brain stem auditory nuclei. It is tonotopically organized and contains a spatial map of the location of sounds.
In sequence, the next segment of the ascending auditory pathway is the medial geniculate nucleus, the thalamic auditory relay nucleus. The medial geniculate nucleus projects to the primary auditory cortex, located within the lateral sulcus on the superior surface of the temporal lobe. The primary auditory cortex contains multiple tonotopically organized territories, all located on Heschl's gyri. The primary cortex forms a central core surrounded multiple secondary auditory areas. Neurons int he primary area are activated by simple tones, whereas those in the secondary areas are better activated by complex sounds. In animals, neurons in these areas are also activated by species-specific calls. the secondary areas are surrounded by several higher-order auditory areas, located on the superior and lateral surfaces of the temporal lobe in superior temporal gyrus and sulcus. This is where several areas are located that are important understanding speech.
The auditory pathways contain decussations and commissures - where axons cross the midline - multiple levels, so that sounds from one ear are processed by both sides of the brain. What is the clinical significance of this bilateral organization of central auditory connections? Unilateral brain damage does not cause deafness in one ear unless the injury destroys the cochlear nuclei or the entering fascicles of the cochlear nerve. Unilateral deafness is thus a sign of injury to the peripheral auditory organ or the cochlear nerve. In addition to being clinically important, the bilateral representation of sounds has a more general significance in providing a mechanism for sound localization and enhancing the detection of sounds through summation of converging inputs.
The Subcortical Nuclei
The Auditory Nervous System
The auditory nervous system consists of two main parts, the ascending and descending system. Ascending the classic lemniscal cochlear nucleus, all of the auditory fibers are interrupted in this nucleus. The dorsal cochlear nucleus, the posterior ventral cochlear nucleus and the anterior ventral cochlear nucleus. Each fiber of the cochlear nerve bifurcates to terminate in the PVCN and AVCN. The fibers that reach the PVCN send collateral fibers to the DCN. In that way, all auditory nerve fibers reach all three divisions of the CN. Neurons of the CN connect to the central nucleus of the inferior colliculus via several fiber tracts that cross the midline and there are also connections that do not cross the midline. Output connects to the thalamic auditory relay nucleus, namely the medial geniculate body which furnishes auditory information to the primary auditory cortex. Primary Auditory cortex is located in Hechel's gyrus in the lateral fissure of the temporal lobe; Broca's Area 41
The connections of the auditory system with the thalamus and cerebral cortex are examined.
Key levels through the brain stem, where auditory information is processed.
Synapses
First synapse: cochlear nucleus (hindbrain)
Establishment of multiple representations of frequency in the cochlear nucleus through branching or primary afferent axons. The neurons of the cochlear nucleus are the last purely monaural nucleus in the auditory system; while monaural neurons occur more rostrally, most neurons are binaural and those driven by the contralateral ear predominate
Second synapse: superior olivary complex (medulla)
Using caution in "numbering" synapses in the central auditory pathway: reciprocity and convergence/divergence makes the establishment of a single linear sequence difficult; the auditory pathway has several channels for analysis. A circuit for establishing binaurality in superior olivary complex neurons
Third synapse: inferior colliculus (midbrain)
Multiple maps of frequency. Establishment of independent maps of aurality: monaural and binaural regions are segregated in the midbrain. The binaural representation is superimposed upon the tonotopic map and most cells respond to both frequency-specific and aural inputs.
Fourth synapse: medial geniculate body (diencephalon)
An important hub for linking auditory and limbic systems
Fifth synapse: auditory cortex (telencephalon)
There are several subdivisions of auditory cortex; their different functions are not understood nor are they essential for all aspects of hearing. Corticofugal descending input to the auditory thalamus, midbrain and other areas
Physiology of Auditory Pathways
Frequency locking and tuning curves of auditory pathways. Phase information is useful primarily at low frequencies. Monaural and binaural representation in the auditory system. Sound localization by interaural intensity cues in localization and interaural time cues in localization and interaural phase differences as cues for frequencies below 8 kHz
Olivocochlear centrifugal system may be a critical link in the control of hair cell length, motility and tuning
relation between olivocochlear system and the outer hair cells. Micromechanical theory of outer hair cell function and the tectorial membrane.
Pathologies & Types of Hearing Loss
Lesions of the auditory system produce level-specific effects; lower lesions are far more serious than forebrain damage
the development of cochlear prostheses is a promising and controversial adjunct to hearing aids
Lesions of the auditory system produce level-specific effects; lower lesions are far more serious than forebrain damage
The development of cochlear prostheses is a promising and controversial adjunct to hearing aids
Pathologies can affect the waveform of the ABR, hearing loss of various kinds could affect the ABR, also depends on three other key factors
Hearing impairments are caused by abnormalities of structure and/or function in the auditory system, which are often called lesions. Using this terminology, a hearing loss may be viewed as one of the manifestations of a lesion somewhere in the ear, as are other symptoms.
Sensorineural Hearing Loss
Acoustic reflex thresholds depend on hearing sensitivity in a rather peculiar way. In this context "hearing sensitivity" represents a continuum going from normal hearing through various magnitudes of sensorineural hearing loss due to cochlear disorders.
It is well established that patients with retrocochlear pathologies have acoustic reflexes that are elevated, often to the extent that the reflex is absent. However, the decision about when and ART is "elevated" must account for the fact that the ARTs depend on the magnitude of the hearing loss in patients who do not have retrocochlear involvement. The 90th percentiles provide us with upper cutoff values for ARTs that meet this need. In fact, many prior inconsistencies about the diagnostic usefulness of the reflex were resolved by the introduction of 90th percentiles that account for the degree of hearing loss.
In practice, the patient's ARTs are compared with the respective 90th percentiles that apply to his hearing thresholds for the frequencies tested. If an ART falls on or below the relevant 90th percentile, then it is considered to be essentially within the normal and/or cochlear distribution. However, ARTs that fall above the applicable 90th percentiles are considered elevated because only a small proportion of normal and/or cochlear-impaired ears have ARTs that are so high. If the abnormally elevated or absent reflexes are not attributable to a conductive disorder, then the patient is considered to be at risk for eighth nerve pathology in the ear that receives the stimulus. In contrast, many patients with functional impairments have ARTs that are below the 10th percentiles.
Abnormal reflex decay means that the response decreases rapidly and is associated with retrocochlear disorders. It is prudent to be suspicious of retrocochlear pathology in the stimulated ear if the reflex response decays by 50% or more within 10 seconds at either 500 Hz and/or 1000 Hz. However, the student should be aware that there are differences in approach regarding whether abnormal decay should occur during the first 5 seconds versus any time during the 10 second test as well as how to interpret reflex decay that occurs at one frequency and/or the other.
We have seen that acoustic reflex abnormalities such as elevated/absent reflexes and/or positive reflex decay are associated with retrocochlear pathologies in the ear receiving the stimulus tone. Testing both ARTs and reflex decay and considering a positive result on either or both tests as the criterion for suspecting retrocochlear pathology has been referred to as acoustic reflexes combined. Acoustic reflex thresholds and decay should be routinely tested because ARC with 90th percentiles has a hit rate of ~85% for retrocochlear pathology and a false positive rate of only 11%.
Sensorineural Hearing Loss
1.Hearing loss can arise in the conductive apparatus before the oval window, when it is known as conductive loss. Alternatively, it may arise in the cochlea or more centrally, when it is known as sensorineural hearing loss. If it arises in the cochlea, it is known as sensorineural hearing loss of cochlear origin.
2. Sensorineural hearing loss of cochlear origin can be caused by ototoxic drugs. Aminoglycoside antibiotics are ototoxic and have been extensively investigated; they damage hair cells, and particularly outer hair cells, in the high-frequency parts of the cochlea. They are likely to cause their damage at least in part by oxidative mechanisms, and antioxidants can provide some protection.
3. Other ototoxic agents include Cisplatin (an anticancer drug), some loop diuretics (bumetanide, furosemide and ethacrynic acid), aspirin (reversibly) and some organic solvents (e.g. styrene and toluene).
4. With acoustic overstimulation, for milder degrees of damage, there is minor disruption of the stereocilia. These degrees of damage are probably reversible. Greater degrees of damage cause loss of the hair cells.
5. Some forms of hearing losses have genetic causes. The most common inherited hearing loss, accounting for some 35% of all congenital (present from birth) hearing losses (depending on the population), is the type known as DFNB1. DFNB1 is caused by a mutation in the gene coding for the gap junction protein connexin-26. Patients with this mutation have problems cycling K+ from the cochlear scala through the cochlear walls back to the stria vascularis. This and many other inherited hearing losses have been invaluable for revealing basic mechanisms of the inner ear.
6. Mutations in the mitochondrial genome may also give rise to hearing loss. These syndromes are maternally inherited, commonly affect multiple organ systems and tend to increase with severity over time.
7. Aging causes sensorineural hearing loss (presbycusis), which can have both cochlear and central components. In the cochlea, vulnerable sites are the hair cells, the nerve and the stria vascularis.
8. Damage to the outer hair cells causes a loss of the active amplification of the traveling wave in the cochlear duct. This leads to a reduction in the magnitude of the traveling wave and to a broadening of the peak of the wave. The changes lead to a loss in the sensitivity of auditory nerve fibers, and a broadening of their tuning curves, that is to a loss of frequency resolution. Damage to the inner hair cells leads to a loss of sensitivity of the auditory nerve fibers without changes in frequency resolution.
9. The psychophysical results fit in with the physiological results. In sensorineural loss of cochlear origin, a loss of frequency resolution is often seen in addition to the loss in sensitivity. The loss in frequency resolution can be seen in widened psychophysical tuning curves and in widened psychophysical filters (critical bands). These factors lead to a particular difficulty in understanding broadband complex sounds, such as speech, and particularly against noisy backgrounds.
10. Some forms of tinnitus, that is those arising in the cochlea from the active mechanical amplification of the traveling wave, may give rise to objectively measurable sound emissions in the ear canal. However, this is unlikely to explain the clinically most important forms of tinnitus. Tinnitus is commonly associated with some degree of hearing loss. This suggests that is arises from a hypersensitivity of the central auditory nervous system when it is deprived of hypersensitivity of the central auditory nervous system when it is deprived of its normal input. It appears that neural activity increases in the auditory pathway, starting at the dorsal cochlear nucleus, and at later stages. The tinnitus may arise from a reduction in inhibition by the neurotransmitters GABA and glycine, and the anti-epileptic drugs vigabatrin and gabapentin that increase GABA inhibition have been reported to reverse tinnitus in experimental animals and some patients. Tinnitus may also arise from down regulation of K+ channels in the nucleus. where there is no effective drug treatment, the behavioral method known as "tinnitus retraining therapy" has proved effective in many patients.
11. With profound sensorineural hearing loss of cochlear origin, hearing can often be restored by a cochlear prosthesis. Current prosthesis consist of multiple electrodes inserted deep into the cochlear duct, where they can stimulate what cells remain of the auditory nerve. Stimuli are pre-processed to pick out the major spectral components of the stimuli, and electrodes at the appropriate positions in the cochlea (depending on the tonotopicity of the cochlea) are stimulated electrically. The amplitude of stimulation depends on the amplitude of the relevant spectral component at each moment, so that the final pattern of stimulation is able to mirror both the place-frequency and temporal aspects of cochlear function. After practice, good communication is achieved with running speech, and 80-90% of open-set sentences are understood correctly.
12. Attempts are being made to replace damaged cells in the inner ear, including the hair cells, cells of the spiral ganglion (auditory nerve) and the stria vascularis. These include stimulating the division of the supporting cells, to produce new hair cels, inoculating the cochlea with a virus engineered to contain a gene Math-1, which encourages the supporting cells to trans-differentiate (transform without undergoing cell division) into hair cells and the injection of stem cells. The transdifferentiation of supporting cells induced by Math-1 has been able to partially restore some hearing in adult animals even after complete loss of all hair cells.However, these methods require some remaining differentiated supporting cells in the organ of Corti, which is not usually the case with advanced sensorineural hearing loss. Stem cells may however form a possible treatment for repair of the auditory nerve.
13. Gene therapy may be possible, where the function of a damaged gene is replaced by an introduced normal copy of the gene. Protection of hair cells may also be possible by inhibiting apoptotic (the death of cells that occurs as a normal and controlled part of an organism's growth or development. Also called programmed cell death) use of antioxidant therapy and manipulations which increase the ear's own biochemical protective mechanisms, for example those arising in the phenomenon know as "sound conditioning" or "toughening", which work via corticosteroid defenses.
cell death: Occurs by necrosis and apoptosis
Necrosis
mutations: 35% due to gene called connexin-26
presbycusis: SNHL due to aging
noise induced oxidative cochlear injury
acoustic trauma: With Acoustic Trauma, frequency of exposure determines frequency range of sensitivity loss
tinnitus:
cochlear implant:
Conductive Hearing Loss
Conductive hearing losses cause acoustic reflexes to be either "elevated" or "absent." By "elevated" we mean that the ART is higher than normal, that is, it takes more intensity to reach the reflex threshold than would have been needed if there was no conductive loss. An "absent" reflex means that a reflex response cannot be obtained, even with the most intense stimulus available (which is usually 125 dB HL on most modern immittance devices). The effects of conductive loss can be summarized by two basic rules:
Auditory Cortex
1.Hearing loss can arise in the conductive apparatus before the oval window, when it is known as conductive loss. Alternatively, it may arise in the cochlea or more centrally, when it is known as sensorineural hearing loss. If it arises in the cochlea, it is known as sensorineural hearing loss of cochlear origin.
2. Sensorineural hearing loss of cochlear origin can be caused by ototoxic drugs. Aminoglycoside antibiotics are ototoxic and have been extensively investigated; they damage hair cells, and particularly outer hair cells, in the high-frequency parts of the cochlea. They are likely to cause their damage at least in part by oxidative mechanisms, and antioxidants can provide some protection.
3. Other ototoxic agents include Cisplatin (an anticancer drug), some loop diuretics (bumetanide, furosemide and ethacrynic acid), aspirin (reversibly) and some organic solvents (e.g. styrene and toluene).
4. With acoustic overstimulation, for milder degrees of damage, there is minor disruption of the stereocilia. These degrees of damage are probably reversible. Greater degrees of damage cause loss of the hair cells.
5. Some forms of hearing losses have genetic causes. The most common inherited hearing loss, accounting for some 35% of all congenital (present from birth) hearing losses (depending on the population), is the type known as DFNB1. DFNB1 is caused by a mutation in the gene coding for the gap junction protein connexin-26. Patients with this mutation have problems cycling K+ from the cochlear scala through the cochlear walls back to the stria vascularis. This and many other inherited hearing losses have been invaluable for revealing basic mechanisms of the inner ear.
6. Mutations in the mitochondrial genome may also give rise to hearing loss. These syndromes are maternally inherited, commonly affect multiple organ systems and tend to increase with severity over time.
7. Aging causes sensorineural hearing loss (presbycusis), which can have both cochlear and central components. In the cochlea, vulnerable sites are the hair cells, the nerve and the stria vascularis.
8. Damage to the outer hair cells causes a loss of the active amplification of the traveling wave in the cochlear duct. This leads to a reduction in the magnitude of the traveling wave and to a broadening of the peak of the wave. The changes lead to a loss in the sensitivity of auditory nerve fibers, and a broadening of their tuning curves, that is to a loss of frequency resolution. Damage to the inner hair cells leads to a loss of sensitivity of the auditory nerve fibers without changes in frequency resolution.
9. The psychophysical results fit in with the physiological results. In sensorineural loss of cochlear origin, a loss of frequency resolution is often seen in addition to the loss in sensitivity. The loss in frequency resolution can be seen in widened psychophysical tuning curves and in widened psychophysical filters (critical bands). These factors lead to a particular difficulty in understanding broadband complex sounds, such as speech, and particularly against noisy backgrounds.
10. Some forms of tinnitus, that is those arising in the cochlea from the active mechanical amplification of the traveling wave, may give rise to objectively measurable sound emissions in the ear canal. However, this is unlikely to explain the clinically most important forms of tinnitus. Tinnitus is commonly associated with some degree of hearing loss. This suggests that is arises from a hypersensitivity of the central auditory nervous system when it is deprived of hypersensitivity of the central auditory nervous system when it is deprived of its normal input. It appears that neural activity increases in the auditory pathway, starting at the dorsal cochlear nucleus, and at later stages. The tinnitus may arise from a reduction in inhibition by the neurotransmitters GABA and glycine, and the anti-epileptic drugs vigabatrin and gabapentin that increase GABA inhibition have been reported to reverse tinnitus in experimental animals and some patients. Tinnitus may also arise from down regulation of K+ channels in the nucleus. where there is no effective drug treatment, the behavioral method known as "tinnitus retraining therapy" has proved effective in many patients.
11. With profound sensorineural hearing loss of cochlear origin, hearing can often be restored by a cochlear prosthesis. Current prosthesis consist of multiple electrodes inserted deep into the cochlear duct, where they can stimulate what cells remain of the auditory nerve. Stimuli are pre-processed to pick out the major spectral components of the stimuli, and electrodes at the appropriate positions in the cochlea (depending on the tonotopicity of the cochlea) are stimulated electrically. The amplitude of stimulation depends on the amplitude of the relevant spectral component at each moment, so that the final pattern of stimulation is able to mirror both the place-frequency and temporal aspects of cochlear function. After practice, good communication is achieved with running speech, and 80-90% of open-set sentences are understood correctly.
12. Attempts are being made to replace damaged cells in the inner ear, including the hair cells, cells of the spiral ganglion (auditory nerve) and the stria vascularis. These include stimulating the division of the supporting cells, to produce new hair cels, inoculating the cochlea with a virus engineered to contain a gene Math-1, which encourages the supporting cells to trans-differentiate (transform without undergoing cell division) into hair cells and the injection of stem cells. The transdifferentiation of supporting cells induced by Math-1 has been able to partially restore some hearing in adult animals even after complete loss of all hair cells.However, these methods require some remaining differentiated supporting cells in the organ of Corti, which is not usually the case with advanced sensorineural hearing loss. Stem cells may however form a possible treatment for repair of the auditory nerve.
13. Gene therapy may be possible, where the function of a damaged gene is replaced by an introduced normal copy of the gene. Protection of hair cells may also be possible by inhibiting apoptotic (the death of cells that occurs as a normal and controlled part of an organism's growth or development. Also called programmed cell death) use of antioxidant therapy and manipulations which increase the ear's own biochemical protective mechanisms, for example those arising in the phenomenon know as "sound conditioning" or "toughening", which work via corticosteroid defenses.
cell death: Occurs by necrosis and apoptosis
Necrosis
mutations: 35% due to gene called connexin-26
presbycusis: SNHL due to aging
noise induced oxidative cochlear injury
acoustic trauma: With Acoustic Trauma, frequency of exposure determines frequency range of sensitivity loss
tinnitus:
cochlear implant:
1.Hearing loss can arise in the conductive apparatus before the oval window, when it is known as conductive loss. Alternatively, it may arise in the cochlea or more centrally, when it is known as sensorineural hearing loss. If it arises in the cochlea, it is known as sensorineural hearing loss of cochlear origin.
2. Sensorineural hearing loss of cochlear origin can be caused by ototoxic drugs. Aminoglycoside antibiotics are ototoxic and have been extensively investigated; they damage hair cells, and particularly outer hair cells, in the high-frequency parts of the cochlea. They are likely to cause their damage at least in part by oxidative mechanisms, and antioxidants can provide some protection.
3. Other ototoxic agents include Cisplatin (an anticancer drug), some loop diuretics (bumetanide, furosemide and ethacrynic acid), aspirin (reversibly) and some organic solvents (e.g. styrene and toluene).
4. With acoustic overstimulation, for milder degrees of damage, there is minor disruption of the stereocilia. These degrees of damage are probably reversible. Greater degrees of damage cause loss of the hair cells.
5. Some forms of hearing losses have genetic causes. The most common inherited hearing loss, accounting for some 35% of all congenital (present from birth) hearing losses (depending on the population), is the type known as DFNB1. DFNB1 is caused by a mutation in the gene coding for the gap junction protein connexin-26. Patients with this mutation have problems cycling K+ from the cochlear scala through the cochlear walls back to the stria vascularis. This and many other inherited hearing losses have been invaluable for revealing basic mechanisms of the inner ear.
6. Mutations in the mitochondrial genome may also give rise to hearing loss. These syndromes are maternally inherited, commonly affect multiple organ systems and tend to increase with severity over time.
7. Aging causes sensorineural hearing loss (presbycusis), which can have both cochlear and central components. In the cochlea, vulnerable sites are the hair cells, the nerve and the stria vascularis.
8. Damage to the outer hair cells causes a loss of the active amplification of the traveling wave in the cochlear duct. This leads to a reduction in the magnitude of the traveling wave and to a broadening of the peak of the wave. The changes lead to a loss in the sensitivity of auditory nerve fibers, and a broadening of their tuning curves, that is to a loss of frequency resolution. Damage to the inner hair cells leads to a loss of sensitivity of the auditory nerve fibers without changes in frequency resolution.
9. The psychophysical results fit in with the physiological results. In sensorineural loss of cochlear origin, a loss of frequency resolution is often seen in addition to the loss in sensitivity. The loss in frequency resolution can be seen in widened psychophysical tuning curves and in widened psychophysical filters (critical bands). These factors lead to a particular difficulty in understanding broadband complex sounds, such as speech, and particularly against noisy backgrounds.
10. Some forms of tinnitus, that is those arising in the cochlea from the active mechanical amplification of the traveling wave, may give rise to objectively measurable sound emissions in the ear canal. However, this is unlikely to explain the clinically most important forms of tinnitus. Tinnitus is commonly associated with some degree of hearing loss. This suggests that is arises from a hypersensitivity of the central auditory nervous system when it is deprived of hypersensitivity of the central auditory nervous system when it is deprived of its normal input. It appears that neural activity increases in the auditory pathway, starting at the dorsal cochlear nucleus, and at later stages. The tinnitus may arise from a reduction in inhibition by the neurotransmitters GABA and glycine, and the anti-epileptic drugs vigabatrin and gabapentin that increase GABA inhibition have been reported to reverse tinnitus in experimental animals and some patients. Tinnitus may also arise from down regulation of K+ channels in the nucleus. where there is no effective drug treatment, the behavioral method known as "tinnitus retraining therapy" has proved effective in many patients.
11. With profound sensorineural hearing loss of cochlear origin, hearing can often be restored by a cochlear prosthesis. Current prosthesis consist of multiple electrodes inserted deep into the cochlear duct, where they can stimulate what cells remain of the auditory nerve. Stimuli are pre-processed to pick out the major spectral components of the stimuli, and electrodes at the appropriate positions in the cochlea (depending on the tonotopicity of the cochlea) are stimulated electrically. The amplitude of stimulation depends on the amplitude of the relevant spectral component at each moment, so that the final pattern of stimulation is able to mirror both the place-frequency and temporal aspects of cochlear function. After practice, good communication is achieved with running speech, and 80-90% of open-set sentences are understood correctly.
12. Attempts are being made to replace damaged cells in the inner ear, including the hair cells, cells of the spiral ganglion (auditory nerve) and the stria vascularis. These include stimulating the division of the supporting cells, to produce new hair cels, inoculating the cochlea with a virus engineered to contain a gene Math-1, which encourages the supporting cells to trans-differentiate (transform without undergoing cell division) into hair cells and the injection of stem cells. The transdifferentiation of supporting cells induced by Math-1 has been able to partially restore some hearing in adult animals even after complete loss of all hair cells.However, these methods require some remaining differentiated supporting cells in the organ of Corti, which is not usually the case with advanced sensorineural hearing loss. Stem cells may however form a possible treatment for repair of the auditory nerve.
13. Gene therapy may be possible, where the function of a damaged gene is replaced by an introduced normal copy of the gene. Protection of hair cells may also be possible by inhibiting apoptotic (the death of cells that occurs as a normal and controlled part of an organism's growth or development. Also called programmed cell death) use of antioxidant therapy and manipulations which increase the ear's own biochemical protective mechanisms, for example those arising in the phenomenon know as "sound conditioning" or "toughening", which work via corticosteroid defenses.
cell death: Occurs by necrosis and apoptosis
Necrosis
mutations: 35% due to gene called connexin-26
presbycusis: SNHL due to aging
noise induced oxidative cochlear injury
acoustic trauma: With Acoustic Trauma, frequency of exposure determines frequency range of sensitivity loss
tinnitus:
cochlear implant:
Sound travels in waves through the atmosphere and are measured in decibels (dB), a logarithmic scale of measurement of sound pressure level. Approximate scale values are as follows: a whisper is about 30 dB, a loud conversation about 50 dB, a jet taking off about 120 dB, a howitzer (artillery firing) is about 150 dB. There are physical dimensions of sound (frequency and wavelength) and psychological dimentions (pitch; a function of experience, culture and musical training) and timber (complex interaction of fundamental and overtone series that modulate one another harmonically to produce unique combinations of overtones). The behavior of sound waves in the air is complex and involves pitch, timbral and echoic cues. Rarefaction and condensation waves use sound emitted by an object to estimate departure and approach. Doppler shifting is the physical correlate of these departing and approaching wavefronts.
The Auditory System Mediates the sense of hearing. The sensory experience described as hearing is as broad as the sound spectrum itself. From signals of impending danger, like a car horn, to the pleasing sounds that fill a concert hall, much of our daily behavior is determined by the sounds around us. The auditory system is also our principal communication portal, allowing us to understand speech. This system, like the somatic sensory and visual systems has a topographic organization determined by the peripheral receptive sheet. And similar to the other systems, the auditory system consists of multiple parallel pathways that engage multiple cortical regions either directly or via complex corticocortical networks. Each auditory pathway is hierarchically organized and has the connections and properties to mediate different aspects of hearing.
The complexity of the auditory pathways derives from the particular properties of natural sounds, with their diverse frequency characteristics, multiple sources of origin, and large dynamic ranges. However, an added measure of complexity is imposed on the human auditory system by the demands of understanding speech. Although the physical characteristics of a spoken word may not be any more complex than many nonlinguistic sounds, the linguistic quality of the stimulus engages unique cortical areas.
Anatomy of the Ear
The ear consists of the outer ear (pinna), middle ear (external auditory meatus) and inner ear (cochlea) where the transduction (processing) of sound occurs. and where the sensory receptors for hearing are located. Sound waves are captured by the outer ear which are funneled into the external auditory meatus which functions as an impedance transformer that facilitates transmission of airborne sound into vibrations of the fluid in the cochlea. The middle ear is filled with air and acts as a cushion behind the tympanic (drum) membrane. Its proper function depends on air pressure in the middle ear cavity being equal to the ambient pressure, facilitated by the opening and closing of the eustachian tube which connects from each ear bilaterally to the posterior roof of mouth. The incus, malleus, and the stapes are the smallest bones in the body and transcribe sound to the cochlear instrument. The stapes performs like a piston with in and out motion that sets the fluid of the cochlea in motion. The skull also conducts sound (bone-conduction) which is useful for low frequencies however, neural transmission representation in the cochlea pushes beyond the physical limits of bone conduction.
Transduction of the auditory pathway
Sound as a stimulus (rarefaction and compression of air and resulting waveforms) activates the tympanic membrane. Mechanical transduction (middle ear ossicles) matches tympanic and hydraulic impedance. Hydraulic transduction (middle ear to endolymph) converts mechanical signals to fluid displacement. Genesis of generator potential (hydraulic to electrical transmission) involves a fluid displacement creating a micromechanical wavefront. The action potential (conversion of generator potential to action potential) is caused when mechanical shearing deforms hair cell cilia. Complex behavior of hair cells reflects mechanical, electrical, neural and muscle-like properties. Relation of basilar membrane structure and hair cell tuning. Hair cell ciliary linkages are spring-like structures signaling length and displacement of hair cells. Electrical tuning of hair cells. Na+ channels in hair cells are localized in specific hair cell membrane subregions.
The process of hearing begins on the body surface, as sounds are conducted by the auricle and external auditory meatus to the tympanic membrane. Mechanical displacement of the tympanic membrane, produced by changes in sound pressure waves, is transmitted to the inner ear by tiny bones termed the middle ear ossicles. The inner ear transduction machinery is located within the temporal bone in a coiled structure called the cochlea. This is the location of the auditory receptors, termed hair cells because they each have a cilia or hair bundle on their apical surface. Each auditory receptor is sensitive to a limited frequency range of sounds. Hair cells in the human cochlea are not bitotically replaced, and their numbers decline throughout life. This reduction results from conditions such as ear infections, exposure to loud sounds or drugs with ototoxic properties, and aging.
The cochlea is remarkably compact and its central projections are strongly divergent. Spiral ganglion cells sit in small clusters embedded in the temporal bone. Some physiological properties of auditory nerve axons; specific afferent frequency tuning, distinct response profiles distinguish VIIIth (vestibulocochlear) nerve fibers, primary-like discharge pattern, build-up units increase the number of spikes/time period, onset units fir most at stimulus onset, other types also exist and this diversity has important theoretical implications for thinking about how hair cells behave and for the possible existence of ganglion cell subtypes.
The Auditory Nerve
The Vestibulocochlear nerve (VIII) is a sensory nerve for hearing and balance and is the only purely sensory nerve subserving two modalities or two separate components. The Vestibular Nuclei receive input from vestibular hair cells via vestibular ganglion cells for equilibrium analysis and project to the forebrain, the oculomotor system and the spinal cord. The cochlear nuclei receive input from spiral ganglion cells that innervate cochlear hair cells. The vestibular component innervates the semicircular canals, saccule, and utricle and mediates balance. The cochlear component innervates the organ of Corti and serves hearing. The Vestibulocochlear VIII nerve leaves the medulla at the cerebellopontine angle to enter the inner ear as two parts, the cochlear division for hearing and the vestibular division for maintenance of equilibrium.
1. The very great majority of fibers present in the auditory nerve innervate inner hair cells.
2. Single fibers of the auditory nerve are always excited by auditory stimuli and never show sustained inhibition to single stimuli.
3. The fibers have lower thresholds to tones of some frequencies than of others. The relation between threshold and stimulus frequency is known as the "tuning curve". Tuning curves show one threshold minimum, at what is known as the characteristic frequency. The threshold rises sharply for frequencies above and below the characteristic frequency. The tuning curve therefore shows a sharp dip in this frequency region. Tuning curves of auditory nerve fibers are similar to the tuning curves of hair cells and of the mechanical response of the basilar membrane.
4. The great majority of (90%) of auditory nerve fibers have minimum thresholds in a 10dB range near the animal's absolute threshold. The others have thresholds spread over a 60dB range above that. The low-threshold fibers have particularly high rates of spontaneous activity in the absence of sound.
5. Fibers show a sigmoidal (S-shaped curve) relation between firing rate and stimulus intensity. Low-threshold fibers also have steeper rate-intensity functions, often going from threshold to maximum rate in 20-30dB (the dynamic range) at any one frequency. Fibers of higher threshold have shallower rate-intensity functions and wider dynamic ranges, up to 60dB or more.
6. The frequency-resolving power of auditory nerve fibers has been measured by a "quality" factor, by analogy with a quality factor for filters. The quality factor is the characteristic frequency divided by the bandwidth of the fiber to tones at an intensity 10dB above the best threshold. This is called "Q10" or "Q10dB". Therefore, fibers with a high Q10 have good frequency selectivity. At any one frequency, different fibers have Q10s in a restricted range. In any one animal, the range of Q10s at one frequency is twofold or less. In cats, the greatest Q10s are reached at around 10kHz, where they have an average value of eight. The Q10s match, to a first approximation at least, the Q10 of the basilar membrane vibration.
7. During tonal stimulation, auditory nerve fibers fire preferentially during one part of the cycle of the stimulating waveform if the frequency of the stimulus is below 4-5kHz. The fibers are excited by deflection of the basilar membrane only in one direction.
8. For fibers with characteristic frequencies below 4-5kHz, clicks preferentially evoke responses at certain intervals after the stimulus. A histogram of action potentials made with respect to time after the stimulus suggests that the fibers are activated by the half cycles of a decaying oscillation of the mechanical resonance on the basilar membrane. The frequency of the oscillation is equal to the characteristic frequency of the fiber.
9. One tone can reduce, or suppress, the response to another, even though single tones are only excitatory. This is called two-tone suppression. The suppression arises from the non-linear properties of the basilar membrane mechanics. Two-tone suppression can also be seen in the basilar membrane mechanics and the responses of inner hair cells. Stimuli other than tones cause suppression too.
10. One stimulus can mask the response to another. Masking mainly occurs because the masking stimulus produces a greater firing rate than the masked stimulus. The other mechanisms of masking are the suppression of the response to one stimulus by another, and adaptation in the fiber produced by an ongoing stimulus.
11. When two-tone stimuli are used, auditory nerve fibers can respond to distortion products as a result of non-linear interactions in the cochlea. One distortion tone, known as the cubic distortion tone, is at a frequency 2f1-f2, where f1 is the lower of the tones presented and f2 the higher.
Auditory nerve fibers do not provide a simple representation of the basilar membrane input/output function. Auditory nerve fibers can show either steeper or shallower rate-level functions after sensorineural hearing loss. Thus compression algorithms that are designed to overcome a reduced dynamic range may need to account for more than reduced basilar membrane compression. this is particularly true in cases of mixed inner hair cell and outer har cell damage where ihc and ohc damage may interact to produce different effects on an responses.
noise induced hearing loss can produce very steep auditory nerve growth at high levels
anatomy of a neuron; dendrites, cell body (soma) and an axon
afferent hair cell; 90-95% of AN synapse w/ inner hair cells each is innervated by multiple; "many to one". 5% outer hair cells synapse afferently termed one to many; one to ten
Types of Fibers: Type 1 - large, myelinated, 95%, inner hair cells; bi-polar cells
Type 2 - small, myelinated and unmyelinated, innervate the ohc; uni-mono
tonotopicity of the auditory nerve: arrangment is a bullseye, with low in the middle and high on the outside
response/characteristic frequency: responsive to single tones and always elicit an excitatory response, never inhibitory
each fiber has a low threshold at one frequency, the characteristic frequency
- characteristic frequency is the frequency that requires the lowest level of stimulation
- also called the best frequency
- align spikes sequences with the onset of stimuli
- divide the stimulus period or observation period T into N
- count the number of spikes
- draw a bar-graph histogram
Tuning Curves: When you record the post-stimulus time histogram, you get a tuning curve
- each fiber has a low threshold at one frequency and threshold and rises rapidly as the frequency is changed
place code & phase locking: if phase-locking is integrated over a population, the code could be reconstructed
- receptors fire in groups, first one, then another, then a third
- the first group of cells to fire are resetting while the second and third groups fire and so on
The Cochlea
A topographic relationship exists between the location of a hair cell in the cochlea and the sound frequency to which the receptor is most sensitive. As discussed below, from the base of the cochlea to the apex, the frequency to which a hair cell is maximally sensitive changes systematically from hight frequencies to low frequencies. This differential frequency sensitivity of hair cells along the length of the cochlea is the basis of the tonotopic organization of auditory reception. Many of the components of the auditory system are tonotopically organized. The topographic relationship between the relationship between the receptor sheet and the central nervous system is similar to that of the somatic sensory and visual systems, where the subcortical nuclei and cortical areas have a somatotopic or retinotopic organization. In each of these cases, the topographic organization of the central representations is determined by the spatial organzation of the peripheral receptive sheet. An important difference exists however. The receptor sheets of the somatic sensory and visual systems are spatial maps representing stimulus location. The cochlea represents the frequency of sounds. Sound source localization is counted by central nervous system auditory neurons located in the spiral ganglion. The central processes of the bipolar neurons from the cochlear division of the vestibulocochlear nerve (cranial nerve VIII). These axons project to the ipsilateral cochlear nuclei, which are located in the rostral medulla.
- THE COCHLEA
- the cochlea is shaped like a snail shell and has three fluid filled compartments; separates sound according to their spectrum and transforms each sound into a neural code in the individual fibers of the auditory portion of the eight cranial nerve
- The cochlea is a hollow, fluid-filled tube (Reissner's membrane) within which the basilar membrane resides
- the basilar membrane supports the hair cells, which are bathed in cochlear endolymph that is essential for their metabolic health and electrophysiological performance
- Reissner's membrane itself is bathed in perilymph, which cushions the membranous labyrinth (which contains the receptors) from direct contact with the bony cochlea
- Tympanic membrane is coupled to the ossicles of the middle ear and it represents the first site of airborne wavefront to tissue contact
- the middle ear is responsible for the transformation of a pressure change to a mechanical displacement
- the ossicles are supported by ligaments that regulate the tightness of coupling between the ossicles and the oval window
- the tightness of this coupling is in part under central neural efferent control and loud noises activate a protective reflex that decouples the ossicles from the oval window and thus desensitizes the system
- The ossicles act as a series of levers to match the impedance of the tympanic membrane which is large and resembles a drum head to the much greater inertia of the perilymph; the actual interface is the bone-fluid junction which is the stapes attachment at the oval window
- The air-borne vibrations displace perilymph, which in turn transmits the waveforms to the endolymph and thus moves the hair cells apical tips against the overlying basilar membrane, creating a receptor potential
- Structure of the basilar membrane is directly related to it's mechano-electrical performance
- the thickness and stiffness of the basilar membrane has a profound micromechanical influence on the tuning of hair cells
- The hair cells
- There are about 20,000 hair cells representing long (outer) and short (inner subtypes)
- the inner hair cells represent only one-fourth of the total and lie in a single row
- however they receive about 95% of the innervation that arises from spiral ganglion cells
- they receive only a fraction of the efferent innervation (the olivocochlear system)
- the outer hair cells form three rows
- few primary afferents end on outer hair cells; there is debate about whether these cells project to the brain
- however, they receive the bulk of the olivocochlear projections
- tuning properties of inner and outer hair cells compared: a paradox of auditory function
The cochlea is a hollow, fluid-filled tube (Reissner's membrane) within which the basilar membrane resides
- the basilar membrane supports the hair cells, which are bathed in cochlear endolymph that is essential for their metabolic health and electrophysiological performance
- Reissner's membrane itself is bathed in perilymph, which cushions the membranous labyrinth (which contains the receptors) from direct contact with the bony cochlea
- the middle ear is responsible for the transformation of a pressure change to a mechanical displacement
- the ossicles are supported by ligaments that regulate the tightness of coupling between the ossicles and the oval window
- the tightness of this coupling is in part under central neural efferent control and loud noises activate a protective reflex that decouples the ossicles from the oval window and thus desensitizes the system
The air-borne vibrations displace perilymph, which in turn transmits the waveforms to the endolymph and thus moves the hair cells apical tips against the overlying basilar membrane, creating a receptor potential
Structure of the basilar membrane is directly related to it's mechano-electrical performance
- the thickness and stiffness of the basilar membrane has a profound micromechanical influence on the tuning of hair cells
The hair cells
- There are about 20,000 hair cells representing long (outer) and short (inner subtypes)
- the inner hair cells represent only one-fourth of the total and lie in a single row
- however they receive about 95% of the innervation that arises from spiral ganglion cells
- they receive only a fraction of the efferent innervation (the olivocochlear system)
- the outer hair cells form three rows
- few primary afferents end on outer hair cells; there is debate about whether these cells project to the brain
- however, they receive the bulk of the olivocochlear projections
- tuning properties of inner and outer hair cells compared: a paradox of auditory function
1. The cochlea is a coiled tube, divided lengthways into three scalae. The three divisions are known as the scala vestibuli, the scala media and the scala tympani. The two outer scalae, the scala vestibuli and the scala tympani contains perilymph, which is like normal extracellular fluid in composition, and is at or near ground potential. The scala media contains endolymph, which is more like intracellular fluid, and has a positive potential (+80mV). The positive potential is generated as a diffusion potential across the intermediate and basal cell layers of the stria vascularis, driven by concentration gradients arising from Na+/K+-ATPase and Na+/2Cl-/K+-ATPase ion pumps in the stria vascularis.
2. The organ of Corti contains the auditory transducers. The organ sits on the basilar membrane dividing the scala medai from the scala tympani. The transducing cells are called hair cells. Hair cells are of two types, known as inner and outer hair cells. They have many hairs, or stereocilia, projecting from their apical (apex) surface. Deflection of the hairs initiates transduction, via a pull on the tip links between the stereocilia, which opens the mechanotransducer channels near the tips of the stereocilia by a direct mechanical action. The molecular identity of the mechanotransducer channels is not known.
3. Deflection of the hairs is produced by deflection of the basilar membrane. The latter occurs as a result of a sound-induced displacement of the cochlear fluids, which interacts with the stiffness of the basilar membrane, to produce a progressive traveling wave on the membrane. The wave passes up the cochlea from base to apex.
4. Traveling waves produced by sounds of high frequency come to a peak near the base of the cochlea. High-frequency sounds are therefore transduced near the base of the cochlea. The traveling wave produced by low-frequency sounds comes to a peak further up the cochlea, and low-frequency sounds are transduced near the apex.
5. The traveling wave has a sharply tuned peak. The sharp peak underlies the sensitivity and frequency selectivity shown by the rest of the auditory system, and indeed the sensitivity and frequency selectivity shown by the whole organism. The sharply tuned peak arises because the outer hair cells, when stimulated by the movement, make an active mechanical response which amplifies the vibration of the basilar membrane as the traveling wave passes through. The traveling wave therefore increases in amplitude as it passes along the cochlear duct, until it dies away abruptly when it reaches a point where the cochlear partition can no longer sustain vibrations of that particular frequency. This point is nearer to the base for high-frequency stimuli and nearer to the apex for low-frequency stimuli.
6. The active amplification has its largest effect at low stimulus intensities. At higher intensities, it makes a smaller contribution. The result is that the basilar membrane moves with a compressive non-linearity, that is the response does not grow as fast as the input. This has an important functional consequence, because it allows the auditory system to discriminate stimuli over a very wide range of stimulus intensities.
7. Deflection of the stereocilia by the traveling wave, by opening and closing ion channels in the stereocilia, modulates the current being driven into the hair cells by the combined effects of the positive endocochlear potential (+80mV) and the negative intracellular potential. The current produces potential changes that can be measured in the hair cells with fine microelectrodes, and grossly in the cochlea with larger electrodes.
8. Inner hair cells have resting potentials of about -45mV. They produce both an a.c. voltage and a steady d.c. depolarization in response to sound. The potentials have sharp tuning curves and amplitude functions similar to those shown by the basilar membrane vibrations. Outer hair cells have resting potentials of about -70mV. They show a.c. potential changes in response to sound and, depending on the circumstances, either no, or a depolarizing, or a hyperpolarizing, d.c. response. Like inner hair cells, they show sharp tuning curves and amplitude functions similar to those shown by basilar membrane vibrations.
9. The potential changes in inner hair cells serve to govern the release of neurotransmitter glutamate, to produce action potentials in the auditory nerve fibers. Outer hair cells amplify the mechanical traveling wave, producing a mechanical motile response in the hair cells. The motile response may arise from one or both of the following: (1) a lengthwise contraction and expansion of the outer hair cell body in response to intracellular depolarization and hyperpolarization or (2) Ca2+ ions entering through the open transducer channels, which may change the conformation of the mechano-transducer apparatus, leading to an output of mechanical energy that can feed back into the mechanical system.
10. The cochlear microphonic is the extracellular correlate of the a.c. current flowing through the hair cells. It is generated predominantly by outer hair cells. The summating potential is primarily the extracellular correlate of the d.c. component of the current flowing through hair cells. Depending on circumstances, it can be recorded as either a positive or negative shift in the scala media and probably receives contributions from both inner and outer hair cells, and possibly from other sources.
11. The massed synchronized activity of auditory nerve fibers, called the compound action potential (CAP) or the N1 and N2 potentials, can be recorded with gross electrodes in response to stimulus onsets.
perilymph and endolymph: perilymph is mostly Na+ and at or near ground potential 0V
endolymph is mostly K+ ions and is at or near +80mV
this creates the endolymphatic potential
stria vascularis produces endolymph
Organ of Corti: approximately 35mm
inner hair cells form a single longitudinal row running along the inner side of the sensory region with respect to the center of the spiral - short and fat
- 95% of auditory nerve fibers
- deflection of stereo cilia open mechanically gated ion channels
- allows for positively charged ions to enter the cell
- influx of ions depolarizes and causes action potentials to fire
- sharpen the frequency resolving power of the cochlea
- is connected to the efferent system
- provide basis for the cochlear amplifier
- sends neural impulses to the brain
- phalangeal (finger) cells next to pillar cells for both to support near the tunnel of corti
- moves up shearing force relative to TM and organ of corti
- results in hair cell deflection
Hair Cell Transduction: when the stereocilia bundle is deflected, the tip link stretches and pulls open the mechanotransducer channel
- allows K+ to flow in and be the vehicle of transduction to drive synaptic transmission
- when the cell is sufficiently depolarized, neurotransmitter is released which causes the auditory nerve to fire
- there is a gradient, the fluid is +80mv, the cell is -45 or -70 which causes a slide of potassium which with calcium forms vesicles that flow across the synaptic cleft to the afferent nerve of the spiral ganglion cells
The Traveling Wave: The response of the basilar membrane underlies the performance of the whole auditory system
- sound pressure waves travel from the tympanic membrane through the ossicles to the stapes footplate
- stapes footplate pushes at the round window
- vibrations from the oval window cause movement of perilymph
- initiates wave of displacement on the basilar membrane
- the wave "travels" from base to apex
- makes a u-turn at the helicotrema and pushes back towards the round window
- the wave pattern and position depend on the frequency
- perilymph is displaced
Determinants of Resonant Frequency:
- Frequency depends on properties of the source of sound
- spring-mass system; mass and stiffness of system (inverse of elasticity)
The Cochlear Amplifier: Prestin!
- amplification provided by the outer hair cells
- amplification is greatest at the lowest sound levels; it gradually diminishes as sound intensity is increased
- amplification can reach as high as 1000 fold
- outer hair cell expansion-contraction cycles in appropriate phase with basilar membrane can boost the latter's amplitude
- direct voltage to force converter
- mediates changes in outer hair cell length, might be responsible for amplification
- this is the stereocilia kick
- large displacement with only a small force
- dancing outer hair cells
ultimately modulating the mechanotransduced action potential
THE COCHLEA IS NOT PASSIVE, IT PRODUCES ENERGY!
boosts up the response to up to 60dB!
Sharp Tuning:
- provides for good hearing sensitivity
- good frequency resolution
- normal growth of loudness
- responsible for otoacoustic emissions
- these changes are non-linear!
- large gain boost at low intensities
EVOKED POTENTIALS:
- Stimulus-evoked recordings of neural activity
- near-field
- close to, in or on the neuronal population
- intracranial
- far-field
- far away from stimulated site
- scalp
reproduces frequency and waveform of a sinusoid perfectly
generated from the outer hair cells
Auditory System Cochlear Nerve
The cochlear nerve (part of the VIII cranial nerve) conducts impulses generated in the organ of Corti to the cochlear nuclei; the central pathways from these nuclei travel to intermediate cell stations of the brain stem and thalamus and on to the cerebral cortex.
The central processes of the spiral ganglion neurons form the cochlear nerve, which leaves the petrous portion of the temporal bone via the internal auditory meatus and runs a short course to the cerebello-pontine angle. On entering the rostral medulla, each axon of the nerve bifurcates, sending a branch to each of the cochlear nuclei, dorsal and ventral. These nuclei are found in the rostral medulla and the caudal pons. The dorsal and ventral nuclei are situated posterolateral and anterolateral to the inferior cerebellar peduncle, respectively.
Efferents from the dorsal cochlear nucleus form the dorsal acoustic stria to cross the pontine tegmentum to a contralateral relay cell complex, the superior olivary nucleus. Giving a collateral to the olivary nucleus, the efferent fibers contribute to the ascending lateral lemniscus. The lateral lemniscus is the major brain stem auditory pathway; it is located in the lateral tegmentum, from the rostral medulla to its termination in the inferior colliculus and the medial geniculate body. Some ascending dorsal cochlear efferents in the lateral lemniscus terminate in the nuclei of the lateral lemniscus, arranged adjacent to and along the extent of the lateral lemniscus; others continue on to the ipsilateral inferior colliculus on that side.
Efferents from the ventral cochlear nucleus may project to the ipsilateral dorsal cochlear nucleus or arc around the inferior cerebellar peduncle. The latter form the intermediate acoustic stria to the ipsilateral or contralateral superior olivary nucleus. These fibers continue on as part of the lateral lemniscus to terminate in the lateral lemniscal nuclei or the ipsilateral inferior colliculus.
Efferents from the ventral cochlear nucleus may also form the ventral acoustic stria or trapezoid body, running transversely through the caudal pons. Collaterals of these efferents terminate in the ipsilateral superior olivary nucleus or in the nuclei of the trapezoid body. The trapezoid nuclei and the neighboring ventral cochlear efferents and their collaterals constitute the trapezoid body complex . The trapezoid body fibers may synapse in the contralateral superior olivary nucleus or bend rostrally to contribute to the lateral lemniscus and ascend to the lateral lemniscal nuclei or inferior colliculus. No fibers from the cochlear nuclei ascend in the ipsilateral lemniscus.
In the midbrain, most of the lateral lemniscal fibers terminate in the inferior colliculus, an important relay center for impulses particularly concerned with auditory reflexes, such as involuntarily jumping in response to a loud noise. From each inferior colliculus, some axons pass to the contralateral colliculus via the commissure of the inferior colliculus; other efferents form the brachium of the inferior colliculus and project to the medial geniculate body of the thalamus.
Neurons of the medial geniculate body send axons to the superior temporal gyrus of the cerebral cortex. These axons form a large bundle called the auditory radiation, composing the sublenticular part of the internal capsule and the lateral part of the corona radiata. The axons of the auditory radiation terminate in area 41 (M; Heschl's gyrus or the primary auditory cortex) on the superior temporal gyrus.
In addition to this extensive afferent pathway, there is also an efferent tract, the olivocochlear bundle. This tract projects to the hair cells of the organ of Corti from contralateral brain stem nuclei, including one of the nuclei of the superior olivary complex. Such a pathway represents an inhibitory feedback mechanism on auditory input; stated in more familiar jargon, we can "turn off" what we don't want to hear.
Destruction of the cochlear nuclei or nerve causes complete deafness on the affected side. Lesions of the lateral lemniscus or area 41 cause minimal bilateral deafness, a condition difficult to detect clinically because of the extensive crossed and uncrossed fibers shown here.
The Subcortical Nuclei
The cochlear nuclei consist of the ventral cochlear nucleus, which has anterior and posterior subdivisions, and the dorsal cochlear nuclus. Neurons in these three components have distinct connections with the rest of the auditory system and give rise to parallel auditory pathways that serve different aspects of hearing. A key function of the anteroventral division of the cochlear nucleus is horizontal localization of sound The posteroventral division contributes to a system of connections that regulate hair cell sensitivity. The two divisions of the ventral cochlear nucleus project to the superior olivary complex, a cluster of nuclei in the caudal pons. Most neurons in the superior olivary complex project via an ascending pathway called the lateral meniscus to the inferior colliculus located in the midbrain. The projection from the ventral cochlear nucleus to the inferior colliculus is bilateral. The dorsal cochlear nucleus is thought to play a role in identifying sound source elevation. It projects directly to the contralateral inferior colliculus, also via the lateral lemniscus. The inferior colliculus is the site of convergence of all lower brain stem auditory nuclei. It is tonotopically organized and contains a spatial map of the location of sounds.
In sequence, the next segment of the ascending auditory pathway is the medial geniculate nucleus, the thalamic auditory relay nucleus. The medial geniculate nucleus projects to the primary auditory cortex, located within the lateral sulcus on the superior surface of the temporal lobe. The primary auditory cortex contains multiple tonotopically organized territories, all located on Heschl's gyri. The primary cortex forms a central core surrounded multiple secondary auditory areas. Neurons int he primary area are activated by simple tones, whereas those in the secondary areas are better activated by complex sounds. In animals, neurons in these areas are also activated by species-specific calls. the secondary areas are surrounded by several higher-order auditory areas, located on the superior and lateral surfaces of the temporal lobe in superior temporal gyrus and sulcus. This is where several areas are located that are important understanding speech.
The auditory pathways contain decussations and commissures - where axons cross the midline - multiple levels, so that sounds from one ear are processed by both sides of the brain. What is the clinical significance of this bilateral organization of central auditory connections? Unilateral brain damage does not cause deafness in one ear unless the injury destroys the cochlear nuclei or the entering fascicles of the cochlear nerve. Unilateral deafness is thus a sign of injury to the peripheral auditory organ or the cochlear nerve. In addition to being clinically important, the bilateral representation of sounds has a more general significance in providing a mechanism for sound localization and enhancing the detection of sounds through summation of converging inputs.
The Subcortical Nuclei
- The analysis of auditory stimuli is progressively undertaken over many stages of the auditory system, where the critical features are progressively extracted to a greater and greater extent as the information is passed up the auditory pathway. The likely reason is that many of the critical features of the acoustic stimulus are intermingled in the stimulus, such that the neural extraction of some of the features degrades the neural representation of other features. This means that extraction of a number of features has to occur in separate parallel streams, with the results being integrated at one or more later stages. When the recombination occurs, the responses to the different features are re-synthesized to form a neural representation of the auditory properties of the object that gave rise to them, that is they begin to define what has been called an "auditory object."
- The auditory system shows an early division into two streams with broadly different functions, forming a stream involved in binaural sound localization and a stream predominantly involved in sound identification.
- At the level of the cochlear nucleus, the binaural sound localization stream involves the anteroventral cochlear nucleus and some cells of the posteroventral cochlear nucleus, which project ventrally over the brainstem in the trapezoid body to the superior olivary complex of both sides. Here, the localization of sound sources in the horizontal direction is extracted on the basis of interaural timing and intensity cues.
- The stream predominantly involved in sound identification includes other cells of the posteroventral cochlear nucleus and dorsal cochlear nucleus and projects directly to the contralateral ventral nucleus of the lateral lemniscus and the contralateral inferior colliculus.
- The cochlear nucleus has three divisions, known as the anteroventral, posteroventral, and dorsal cochlear nuclei. Each division of the nucleus is tonotopically organized, so that the best frequencies of the neurons make a spatially ordered map in each division. In the anteroventral division, the neuronal responses are similar to those of auditory nerve fibers, with simple tuning curves, no inhibitory sidebands and monotonic rate-intensity functions. Globular and spherical bushy predominant, each receiving synaptic terminals of the auditory nerve fibers in the form of a few large end bulbs of Held, giving fast and secure activation of the neurons. They project to the superior olivary complex via the ventral, binaural sound localization stream.
- In the posteroventral cochlear nucleus, as well as globular bushy cells, there are octopus cells, which have broad tuning curves and fast temporal responses. Octopus cells can follow the temporal variations in broadband stimuli up to high rates.
- The cells project via the dorsal predominantly sound identification pathway, primarily to the contralateral cochlear nucleus of the lateral lemniscus.
- The posteroventral cochlear nucleus also contains stellate cells (also present in the anteroventral nucleus). Stellate cells (also known as multipolar cells) are of two types, the more numerous T-stellate cells and the less numerous D-stellate cells.
- T-stellate cells project bilaterally to the inferior colliculus and to the ventral nucleus of the lateral lemniscus as well as to the cochlear nucleus and superior olive.
- D-stellate cells send inhibitory collaterals widely within the cochlear nuclei.
- The dorsal cochlear nucleus has multiple cell types, the major output cells being the fusiform cells. Cells of the dorsal nucleus tend to have very complex tuning curves, strong bands of inhibition and non-monotonic rate-intensity functions. Cells of the dorsal nucleus, as well as projecting within the nucleus, project via the dorsal stream which is predominantly involved in sound identification to the contralateral inferior colliculus. Some of the cells of the dorsal nucleus respond well to spectral notches and may be involved in judging the elevation of sound sources based on spectral cues introduced by the pinna. Many cells in the cochlear nucleus emphasize spectral contrast and temporal fluctuations in the stimulus.
- The superior olivary complex receives an input from the anteroventral and posteroventral cochlear nuclei. The superior olivary complex has several component nuclei. The largest component nucleus, the lateral superior olive (LSO), receives an input from both sides , the ipsilateral being excitatory and the contralateral input inhibitory, via a synapse in the medial nucleus of the trapezoid body.
- The nucleus is therefore responsive to differences in interaural sound intensity. It is mainly a high-frequency nucleus and uses the differences in interaural sound intensity and time of arrival of transients in the stimulus to code the direction of mainly high-frequency sound sources, being most strongly driven by sound sources on the ipsilateral side of the head.
- Another component nucleus, the medial superior olive, is mainly a low-frequency nucleus. It is responsive to differences in interaural timing and uses these to code the direction of low-frequency sound sources in space, being most strongly driven by sound sources on the contralateral side of the head.
- The projections from the superior olivary complex and some of the projections from the cochlear nucleus, travel to the inferior colliculus in the tract known as the lateral lemniscus. Some of the fibers in the tract synapse in the dorsal and ventral nuclei of the lateral lemniscus. The ventral nucleus of the lateral lemniscus (VNLL) is part of the sound identification stream. Its anatomical structure has a complex patchiness which suggest that it contributes complex cross-frequency interactions to the inferior colliculus. The dorsal nucleus of the lateral lemniscus (DNLL) is part of the sound localization stream and enhances the lateralization of stimuli in the auditory system. The fibers within the lateral lemniscus and from the VNLL and DNLL project to the inferior colliculus in such a way that neurons in the colliculus, and in all stages in the auditory system thereafter, predominantly represent sound sources on the contralateral side of the head.
- The inferior colliculus marks a jump in the complexity of the responses. Here, the results of the different analysis that were undertaken in separate streams at the lower levels of the auditory system are combined to give responses that begin to define an auditory object. The inferior colliculus has three main divisions, namely a central nucleus (the specific, core or lemniscal auditory relay) and two non-specific, diffuse belt or extra-lemniscal nuclei, called the dorsal cortex and external nucleus.
- The central nucleus has a pronounced laminar structure, with the lamina corresponding to isofrequency planes, otherwise known as frequency-band lamina. The nucleus receives a multiplicity of excitatory and inhibitory inputs from the lower auditory nuclei. A great variety of response types are seen in the central nucleus , with some very sharp and some very wide tuning curves.
- Many cells show enhanced responses to temporal fluctuations and most neurons are responsive to the location of sound sources. There is evidence that some of these properties are distributed differentially and in patches across each frequency-band lamina.
- Neurons in the less specific dorsal cortex and external nucleus are often broadly tuned and tend to habituate rapidly. Neurons in the dorsal cortex show a particularly strong response to novel stimuli and so may have a role in processing novel stimuli.
- Neurons in the external nucleus commonly respond to the directions of sound sources and often have multimodal responses (e.g. to auditory and somatosensory stimuli). The external nucleus is therefore likely to be an auditory and somatosensory integrative area, governing among other things spatial responses to sound.
- The medial geniculate body is the specific thalamic relay of the auditory system, receiving afferents from the inferior colliculus and projecting to the auditory cortex. It has three divisions, namely the ventral, dorsal and medial divisions.
- The ventral division is the specific, core or lemniscal auditory relay and projects to the primary auditory cortex. It has a laminar structure, where individual adjacent lamina are likely to be organized together into functional units called "slabs." The ventral division further sharpens frequency resolution and in species with auditory specializations perform further analysis related to those specializations. It also has heavy reciprocal connections with the auditory cortex and must be seen as a functional unit with the cortex.
- The medial and dorsal divisions of the medial geniculate body form part of the non-specific, diffuse, belt or extra-lemniscal auditory system. They project widely to the areas of cortex surrounding the primary auditory cortex. They have multimodal interactions (i.e. visual and somatosensory as well as auditory responses) and have responses that are modifiable as a result of learning. The dorsal division in particular seems to be involved in the response to novel stimuli. The medial division has a close association with the amygdala and is likely to be involved in the learning of responses to fear-evoking stimuli.
- Brainstem auditory reflexes include
- the middle ear muscle reflex , which involves a reflex arc of two to four neurons, starting at the cochlear nucleus and going to the motor nuclei of the facial and trigeminal nerves, either directly or via the superior olive
- acoustic startle, which is a reflexive contraction of the muscles of the head and neck and which uses a projection from the ventral cochlear nucleus to a nucleus in the reticular formation, the nucleus reticularis pontis caudalis
- reflexive orientation to auditory stimuli, which involves the external nucleus of the inferior colliculus and the superior colliculus
- audiogenic seizures which depend on the inferior colliculus especially its external nucleus and dorsal cortex. The seizures arise from a hypersensitivity of the auditory system, probably in these nuclei, that develops in certain susceptible strains of animals in response to early auditory deprivation.
The Auditory Nervous System
The auditory nervous system consists of two main parts, the ascending and descending system. Ascending the classic lemniscal cochlear nucleus, all of the auditory fibers are interrupted in this nucleus. The dorsal cochlear nucleus, the posterior ventral cochlear nucleus and the anterior ventral cochlear nucleus. Each fiber of the cochlear nerve bifurcates to terminate in the PVCN and AVCN. The fibers that reach the PVCN send collateral fibers to the DCN. In that way, all auditory nerve fibers reach all three divisions of the CN. Neurons of the CN connect to the central nucleus of the inferior colliculus via several fiber tracts that cross the midline and there are also connections that do not cross the midline. Output connects to the thalamic auditory relay nucleus, namely the medial geniculate body which furnishes auditory information to the primary auditory cortex. Primary Auditory cortex is located in Hechel's gyrus in the lateral fissure of the temporal lobe; Broca's Area 41
The connections of the auditory system with the thalamus and cerebral cortex are examined.
Key levels through the brain stem, where auditory information is processed.
Synapses
First synapse: cochlear nucleus (hindbrain)
Establishment of multiple representations of frequency in the cochlear nucleus through branching or primary afferent axons. The neurons of the cochlear nucleus are the last purely monaural nucleus in the auditory system; while monaural neurons occur more rostrally, most neurons are binaural and those driven by the contralateral ear predominate
Second synapse: superior olivary complex (medulla)
Using caution in "numbering" synapses in the central auditory pathway: reciprocity and convergence/divergence makes the establishment of a single linear sequence difficult; the auditory pathway has several channels for analysis. A circuit for establishing binaurality in superior olivary complex neurons
Third synapse: inferior colliculus (midbrain)
Multiple maps of frequency. Establishment of independent maps of aurality: monaural and binaural regions are segregated in the midbrain. The binaural representation is superimposed upon the tonotopic map and most cells respond to both frequency-specific and aural inputs.
Fourth synapse: medial geniculate body (diencephalon)
An important hub for linking auditory and limbic systems
Fifth synapse: auditory cortex (telencephalon)
There are several subdivisions of auditory cortex; their different functions are not understood nor are they essential for all aspects of hearing. Corticofugal descending input to the auditory thalamus, midbrain and other areas
Physiology of Auditory Pathways
Frequency locking and tuning curves of auditory pathways. Phase information is useful primarily at low frequencies. Monaural and binaural representation in the auditory system. Sound localization by interaural intensity cues in localization and interaural time cues in localization and interaural phase differences as cues for frequencies below 8 kHz
Olivocochlear centrifugal system may be a critical link in the control of hair cell length, motility and tuning
relation between olivocochlear system and the outer hair cells. Micromechanical theory of outer hair cell function and the tectorial membrane.
Pathologies & Types of Hearing Loss
Lesions of the auditory system produce level-specific effects; lower lesions are far more serious than forebrain damage
the development of cochlear prostheses is a promising and controversial adjunct to hearing aids
Lesions of the auditory system produce level-specific effects; lower lesions are far more serious than forebrain damage
The development of cochlear prostheses is a promising and controversial adjunct to hearing aids
Pathologies can affect the waveform of the ABR, hearing loss of various kinds could affect the ABR, also depends on three other key factors
Hearing impairments are caused by abnormalities of structure and/or function in the auditory system, which are often called lesions. Using this terminology, a hearing loss may be viewed as one of the manifestations of a lesion somewhere in the ear, as are other symptoms.
Sensorineural Hearing Loss
Acoustic reflex thresholds depend on hearing sensitivity in a rather peculiar way. In this context "hearing sensitivity" represents a continuum going from normal hearing through various magnitudes of sensorineural hearing loss due to cochlear disorders.
It is well established that patients with retrocochlear pathologies have acoustic reflexes that are elevated, often to the extent that the reflex is absent. However, the decision about when and ART is "elevated" must account for the fact that the ARTs depend on the magnitude of the hearing loss in patients who do not have retrocochlear involvement. The 90th percentiles provide us with upper cutoff values for ARTs that meet this need. In fact, many prior inconsistencies about the diagnostic usefulness of the reflex were resolved by the introduction of 90th percentiles that account for the degree of hearing loss.
In practice, the patient's ARTs are compared with the respective 90th percentiles that apply to his hearing thresholds for the frequencies tested. If an ART falls on or below the relevant 90th percentile, then it is considered to be essentially within the normal and/or cochlear distribution. However, ARTs that fall above the applicable 90th percentiles are considered elevated because only a small proportion of normal and/or cochlear-impaired ears have ARTs that are so high. If the abnormally elevated or absent reflexes are not attributable to a conductive disorder, then the patient is considered to be at risk for eighth nerve pathology in the ear that receives the stimulus. In contrast, many patients with functional impairments have ARTs that are below the 10th percentiles.
Abnormal reflex decay means that the response decreases rapidly and is associated with retrocochlear disorders. It is prudent to be suspicious of retrocochlear pathology in the stimulated ear if the reflex response decays by 50% or more within 10 seconds at either 500 Hz and/or 1000 Hz. However, the student should be aware that there are differences in approach regarding whether abnormal decay should occur during the first 5 seconds versus any time during the 10 second test as well as how to interpret reflex decay that occurs at one frequency and/or the other.
We have seen that acoustic reflex abnormalities such as elevated/absent reflexes and/or positive reflex decay are associated with retrocochlear pathologies in the ear receiving the stimulus tone. Testing both ARTs and reflex decay and considering a positive result on either or both tests as the criterion for suspecting retrocochlear pathology has been referred to as acoustic reflexes combined. Acoustic reflex thresholds and decay should be routinely tested because ARC with 90th percentiles has a hit rate of ~85% for retrocochlear pathology and a false positive rate of only 11%.
Sensorineural Hearing Loss
1.Hearing loss can arise in the conductive apparatus before the oval window, when it is known as conductive loss. Alternatively, it may arise in the cochlea or more centrally, when it is known as sensorineural hearing loss. If it arises in the cochlea, it is known as sensorineural hearing loss of cochlear origin.
2. Sensorineural hearing loss of cochlear origin can be caused by ototoxic drugs. Aminoglycoside antibiotics are ototoxic and have been extensively investigated; they damage hair cells, and particularly outer hair cells, in the high-frequency parts of the cochlea. They are likely to cause their damage at least in part by oxidative mechanisms, and antioxidants can provide some protection.
3. Other ototoxic agents include Cisplatin (an anticancer drug), some loop diuretics (bumetanide, furosemide and ethacrynic acid), aspirin (reversibly) and some organic solvents (e.g. styrene and toluene).
4. With acoustic overstimulation, for milder degrees of damage, there is minor disruption of the stereocilia. These degrees of damage are probably reversible. Greater degrees of damage cause loss of the hair cells.
5. Some forms of hearing losses have genetic causes. The most common inherited hearing loss, accounting for some 35% of all congenital (present from birth) hearing losses (depending on the population), is the type known as DFNB1. DFNB1 is caused by a mutation in the gene coding for the gap junction protein connexin-26. Patients with this mutation have problems cycling K+ from the cochlear scala through the cochlear walls back to the stria vascularis. This and many other inherited hearing losses have been invaluable for revealing basic mechanisms of the inner ear.
6. Mutations in the mitochondrial genome may also give rise to hearing loss. These syndromes are maternally inherited, commonly affect multiple organ systems and tend to increase with severity over time.
7. Aging causes sensorineural hearing loss (presbycusis), which can have both cochlear and central components. In the cochlea, vulnerable sites are the hair cells, the nerve and the stria vascularis.
8. Damage to the outer hair cells causes a loss of the active amplification of the traveling wave in the cochlear duct. This leads to a reduction in the magnitude of the traveling wave and to a broadening of the peak of the wave. The changes lead to a loss in the sensitivity of auditory nerve fibers, and a broadening of their tuning curves, that is to a loss of frequency resolution. Damage to the inner hair cells leads to a loss of sensitivity of the auditory nerve fibers without changes in frequency resolution.
9. The psychophysical results fit in with the physiological results. In sensorineural loss of cochlear origin, a loss of frequency resolution is often seen in addition to the loss in sensitivity. The loss in frequency resolution can be seen in widened psychophysical tuning curves and in widened psychophysical filters (critical bands). These factors lead to a particular difficulty in understanding broadband complex sounds, such as speech, and particularly against noisy backgrounds.
10. Some forms of tinnitus, that is those arising in the cochlea from the active mechanical amplification of the traveling wave, may give rise to objectively measurable sound emissions in the ear canal. However, this is unlikely to explain the clinically most important forms of tinnitus. Tinnitus is commonly associated with some degree of hearing loss. This suggests that is arises from a hypersensitivity of the central auditory nervous system when it is deprived of hypersensitivity of the central auditory nervous system when it is deprived of its normal input. It appears that neural activity increases in the auditory pathway, starting at the dorsal cochlear nucleus, and at later stages. The tinnitus may arise from a reduction in inhibition by the neurotransmitters GABA and glycine, and the anti-epileptic drugs vigabatrin and gabapentin that increase GABA inhibition have been reported to reverse tinnitus in experimental animals and some patients. Tinnitus may also arise from down regulation of K+ channels in the nucleus. where there is no effective drug treatment, the behavioral method known as "tinnitus retraining therapy" has proved effective in many patients.
11. With profound sensorineural hearing loss of cochlear origin, hearing can often be restored by a cochlear prosthesis. Current prosthesis consist of multiple electrodes inserted deep into the cochlear duct, where they can stimulate what cells remain of the auditory nerve. Stimuli are pre-processed to pick out the major spectral components of the stimuli, and electrodes at the appropriate positions in the cochlea (depending on the tonotopicity of the cochlea) are stimulated electrically. The amplitude of stimulation depends on the amplitude of the relevant spectral component at each moment, so that the final pattern of stimulation is able to mirror both the place-frequency and temporal aspects of cochlear function. After practice, good communication is achieved with running speech, and 80-90% of open-set sentences are understood correctly.
12. Attempts are being made to replace damaged cells in the inner ear, including the hair cells, cells of the spiral ganglion (auditory nerve) and the stria vascularis. These include stimulating the division of the supporting cells, to produce new hair cels, inoculating the cochlea with a virus engineered to contain a gene Math-1, which encourages the supporting cells to trans-differentiate (transform without undergoing cell division) into hair cells and the injection of stem cells. The transdifferentiation of supporting cells induced by Math-1 has been able to partially restore some hearing in adult animals even after complete loss of all hair cells.However, these methods require some remaining differentiated supporting cells in the organ of Corti, which is not usually the case with advanced sensorineural hearing loss. Stem cells may however form a possible treatment for repair of the auditory nerve.
13. Gene therapy may be possible, where the function of a damaged gene is replaced by an introduced normal copy of the gene. Protection of hair cells may also be possible by inhibiting apoptotic (the death of cells that occurs as a normal and controlled part of an organism's growth or development. Also called programmed cell death) use of antioxidant therapy and manipulations which increase the ear's own biochemical protective mechanisms, for example those arising in the phenomenon know as "sound conditioning" or "toughening", which work via corticosteroid defenses.
- Benign neuroma or schwannoma
- within cochlea
- acoustic trauma
- infections
- congenital anomalies
- age-related
- ototoxic agents; High Frequencies are lost first
- aminoglycosides
- antibiotics; gentamycin, kanamycin
- aspirin may counteract gentamycin
- taken up by hair cells
- cause damage via free radicals
- antibiotics; gentamycin, kanamycin
- loop diuretics
- salicylates
- aminoglycosides
- acoustic overstimulation
- genetic causes
cell death: Occurs by necrosis and apoptosis
Necrosis
- blebs form
- blebs fuse
- blebs rupture
- blebs form
- nucleus breaks apart
- cells breaks into bodies
mutations: 35% due to gene called connexin-26
- allow ions to pass between cells
- gap junctions in the stria vascularis
- causes severe, early onset hearing loss
presbycusis: SNHL due to aging
- sensory pattern associated with hair cell changes
- strial pattern associated with changes in the stria vascularis
- structural patterns with high energy consumption
- mitochondrial dysfunction
- HIGH FREQUENCIES ARE LOST BILATERALLY
noise induced oxidative cochlear injury
acoustic trauma: With Acoustic Trauma, frequency of exposure determines frequency range of sensitivity loss
- sudden, permanent SNHL caused by single exposure to an intense sound
- impulse sound, 130-140dB
- notch at 4kHz
tinnitus:
- auditory sensation in the absence of auditory input
- can be induced by ototoxic drugs
- thought that inhibition is decreased
- disinhibition
- GABA is inhibitory (glutamate is excitatory)
- perhaps reduction of GABA in certain structures may give rise to tinnitus
cochlear implant:
- microphone
- speech processor
- transmitter/receiver/stimulator
- electrodes
- pattern of stimulation mimics phase-locking
Conductive Hearing Loss
Conductive hearing losses cause acoustic reflexes to be either "elevated" or "absent." By "elevated" we mean that the ART is higher than normal, that is, it takes more intensity to reach the reflex threshold than would have been needed if there was no conductive loss. An "absent" reflex means that a reflex response cannot be obtained, even with the most intense stimulus available (which is usually 125 dB HL on most modern immittance devices). The effects of conductive loss can be summarized by two basic rules:
- Probe-ear rule - The presence of conductive pathology in the probe ear causes the acoustic reflex to be absent. Here, even though the stapedius muscle itself may actually be contracting the presence of the pathology prevents us from being able to register any change in acoustic admittance that can be picked up by the probe tip.
- Stimulus-ear rule - A conductive loss in the stimulus ear causes the ART to be elevated by the amount of the conductive impairment. This occurs because the amount of the stimulus that actually reaches the cochlea will be reduced by the amount of the air-bone-gap. For example, suppose an otherwise normal patient develops otitis media with a 25dB air-bone-gap. The 25-dB-air-bone-gap causes the signal reaching the cochlea to be 25 dB weaker than the level presented from the earphone. If her ART would normally have been 85 dB HL (without the conductive loss) then the stimulus would now have to be raised by 25 dB to 110 dB HL to reach her cochlea at 85 dB HL. Hence, the ART would now be elevated to 110 dB HL. In addition, if the air-bone-gap is large enough, then the ART will be elevated so much that the reflex will be absent. This occurs because the highest available stimulus level cannot overcome the size of the air-bone-gap and still deliver a large enough signal to the cochlea.
Auditory Cortex
- Auditory cortex consists of a core surrounded by a belt and a parabelt. The core contains multiple area, including AI. The core receives its major input from the specific or lemniscal division of the thalamic nucleus, namely the ventral division of the medial geniculate body that projects to the auditory cortex. The belt and parabelt areas predominantly receive their inputs from other divisions of the medial geniculate body. The belt also receives intracortical projections from the core, and the parabelt regions receive projections from the belt.
- Auditory cortical areas are defined by the following criteria: (1) cell types and histological appearance (2) connections with the thalamus (3) pattern of staining for cytochrome oxidase, acetylcholinesterase and parvalbumin and (5) the existence of separate tonotopic progressions in some areas.
- In human beings, the primary auditory cortex is situated in the posterior and medial part of Heschl's gyrus on the superior temporal plane on the upper surface of the temporal lobe, deep in the lateral (Sylvian) sulcus. Belt and parabelt areas surround it rostrally, caudally and laterally.
- Primary cortical areas and some of the belt areas are tonotopically organized., that is the characteristic frequencies of neurons change in a progressive manner across the cortex. Lines joining neurons of similar characteristic frequency run orthogonal (at right angles) to the frequency progression and make what are called frequency band strips. Cells whose responses are dominated by one ear or the other are grouped in patches on the surface of the cortex. Other groupings of cells, not related to the above patterns, can be found when cells are analyzed in terms of latency, sharpness of tuning, threshold or sensitivity to frequency modulation. The significance of these groupings is not known.
- Single neurons in the core area show many different shapes of tuning curves in response to pure tones. Some neurons have sharp tuning curves with single frequency region of maximum sensitivity. Others have very broad tuning curves, or multi-peaked tuning curves with two or more frequency regions of maximum sensitivity. Stimuli in the different regions of maximum sensitivity can be facilitory, so that the neurons are particularly responsive to certain complex stimuli with a rich harmonic structure, such as vocalizations. In other cases, although a single excitatory peak is visible in the tuning curve in response to single tones, multiple bands of excitation or inhibition are revealed when further tones are superimposed.
- Outside AI, in other parts of the core and in the belt areas, neurons seem particularly responsive to complex stimuli, including frequency and amplitude modulated tones, with different cortical areas being responsive to different modulation rates.
- Sound location is represented in the auditory cortex, and unilateral lesions in the auditory cortex interfere with behavioral sound localization on the contralateral side. In the cat, local reversible cooling shows that AI, another core area (PAF) and a belt area (AES) are all essential for behavioral sound localization. In these areas, as well as other cortical areas, a high proportion of neurons are sensitive to the localization of sound sources and use interaural intensity cues and or timing as cues.
- Micro-electrode studies in non-human primates and fMRI in human and non-human primates, suggest that after the core areas, spatial information is processed more caudally and dorsally in the cortex, in what has been called the "where" or "do" stream. This includes the posterior temporal gyrus, the inferior parietal cortex and a more anteriorly situated area, near the superior frontal sulcus. On the other hand, complex stimuli particularly activate areas anterior to the auditory core, including in human the planum polare and area around the inferior frontal gyrus. These areas are part of the more rostral "what" stream. However, there is some crossover and some "what" information is also represented in the other stream.
- It is suggested that one role of the auditory cortex in perception, among others, is that of representing auditory objects. Different objects are likely to be represented in overlapping patterns of activity spread over many neurons. The pattern of responsiveness of the cortex at any one time reflects the current behavioral state of the animal, with the neuronal responses modifiable such that a larger proportion of cortex is devoted to stimuli of particular current significance.
1.Hearing loss can arise in the conductive apparatus before the oval window, when it is known as conductive loss. Alternatively, it may arise in the cochlea or more centrally, when it is known as sensorineural hearing loss. If it arises in the cochlea, it is known as sensorineural hearing loss of cochlear origin.
2. Sensorineural hearing loss of cochlear origin can be caused by ototoxic drugs. Aminoglycoside antibiotics are ototoxic and have been extensively investigated; they damage hair cells, and particularly outer hair cells, in the high-frequency parts of the cochlea. They are likely to cause their damage at least in part by oxidative mechanisms, and antioxidants can provide some protection.
3. Other ototoxic agents include Cisplatin (an anticancer drug), some loop diuretics (bumetanide, furosemide and ethacrynic acid), aspirin (reversibly) and some organic solvents (e.g. styrene and toluene).
4. With acoustic overstimulation, for milder degrees of damage, there is minor disruption of the stereocilia. These degrees of damage are probably reversible. Greater degrees of damage cause loss of the hair cells.
5. Some forms of hearing losses have genetic causes. The most common inherited hearing loss, accounting for some 35% of all congenital (present from birth) hearing losses (depending on the population), is the type known as DFNB1. DFNB1 is caused by a mutation in the gene coding for the gap junction protein connexin-26. Patients with this mutation have problems cycling K+ from the cochlear scala through the cochlear walls back to the stria vascularis. This and many other inherited hearing losses have been invaluable for revealing basic mechanisms of the inner ear.
6. Mutations in the mitochondrial genome may also give rise to hearing loss. These syndromes are maternally inherited, commonly affect multiple organ systems and tend to increase with severity over time.
7. Aging causes sensorineural hearing loss (presbycusis), which can have both cochlear and central components. In the cochlea, vulnerable sites are the hair cells, the nerve and the stria vascularis.
8. Damage to the outer hair cells causes a loss of the active amplification of the traveling wave in the cochlear duct. This leads to a reduction in the magnitude of the traveling wave and to a broadening of the peak of the wave. The changes lead to a loss in the sensitivity of auditory nerve fibers, and a broadening of their tuning curves, that is to a loss of frequency resolution. Damage to the inner hair cells leads to a loss of sensitivity of the auditory nerve fibers without changes in frequency resolution.
9. The psychophysical results fit in with the physiological results. In sensorineural loss of cochlear origin, a loss of frequency resolution is often seen in addition to the loss in sensitivity. The loss in frequency resolution can be seen in widened psychophysical tuning curves and in widened psychophysical filters (critical bands). These factors lead to a particular difficulty in understanding broadband complex sounds, such as speech, and particularly against noisy backgrounds.
10. Some forms of tinnitus, that is those arising in the cochlea from the active mechanical amplification of the traveling wave, may give rise to objectively measurable sound emissions in the ear canal. However, this is unlikely to explain the clinically most important forms of tinnitus. Tinnitus is commonly associated with some degree of hearing loss. This suggests that is arises from a hypersensitivity of the central auditory nervous system when it is deprived of hypersensitivity of the central auditory nervous system when it is deprived of its normal input. It appears that neural activity increases in the auditory pathway, starting at the dorsal cochlear nucleus, and at later stages. The tinnitus may arise from a reduction in inhibition by the neurotransmitters GABA and glycine, and the anti-epileptic drugs vigabatrin and gabapentin that increase GABA inhibition have been reported to reverse tinnitus in experimental animals and some patients. Tinnitus may also arise from down regulation of K+ channels in the nucleus. where there is no effective drug treatment, the behavioral method known as "tinnitus retraining therapy" has proved effective in many patients.
11. With profound sensorineural hearing loss of cochlear origin, hearing can often be restored by a cochlear prosthesis. Current prosthesis consist of multiple electrodes inserted deep into the cochlear duct, where they can stimulate what cells remain of the auditory nerve. Stimuli are pre-processed to pick out the major spectral components of the stimuli, and electrodes at the appropriate positions in the cochlea (depending on the tonotopicity of the cochlea) are stimulated electrically. The amplitude of stimulation depends on the amplitude of the relevant spectral component at each moment, so that the final pattern of stimulation is able to mirror both the place-frequency and temporal aspects of cochlear function. After practice, good communication is achieved with running speech, and 80-90% of open-set sentences are understood correctly.
12. Attempts are being made to replace damaged cells in the inner ear, including the hair cells, cells of the spiral ganglion (auditory nerve) and the stria vascularis. These include stimulating the division of the supporting cells, to produce new hair cels, inoculating the cochlea with a virus engineered to contain a gene Math-1, which encourages the supporting cells to trans-differentiate (transform without undergoing cell division) into hair cells and the injection of stem cells. The transdifferentiation of supporting cells induced by Math-1 has been able to partially restore some hearing in adult animals even after complete loss of all hair cells.However, these methods require some remaining differentiated supporting cells in the organ of Corti, which is not usually the case with advanced sensorineural hearing loss. Stem cells may however form a possible treatment for repair of the auditory nerve.
13. Gene therapy may be possible, where the function of a damaged gene is replaced by an introduced normal copy of the gene. Protection of hair cells may also be possible by inhibiting apoptotic (the death of cells that occurs as a normal and controlled part of an organism's growth or development. Also called programmed cell death) use of antioxidant therapy and manipulations which increase the ear's own biochemical protective mechanisms, for example those arising in the phenomenon know as "sound conditioning" or "toughening", which work via corticosteroid defenses.
- Benign neuroma or schwannoma
- within cochlea
- acoustic trauma
- infections
- congenital anomalies
- age-related
- ototoxic agents; High Frequencies are lost first
- aminoglycosides
- antibiotics; gentamycin, kanamycin
- aspirin may counteract gentamycin
- taken up by hair cells
- cause damage via free radicals
- antibiotics; gentamycin, kanamycin
- loop diuretics
- salicylates
- aminoglycosides
- acoustic overstimulation
- genetic causes
cell death: Occurs by necrosis and apoptosis
Necrosis
- blebs form
- blebs fuse
- blebs rupture
- blebs form
- nucleus breaks apart
- cells breaks into bodies
mutations: 35% due to gene called connexin-26
- allow ions to pass between cells
- gap junctions in the stria vascularis
- causes severe, early onset hearing loss
presbycusis: SNHL due to aging
- sensory pattern associated with hair cell changes
- strial pattern associated with changes in the stria vascularis
- structural patterns with high energy consumption
- mitochondrial dysfunction
- HIGH FREQUENCIES ARE LOST BILATERALLY
noise induced oxidative cochlear injury
acoustic trauma: With Acoustic Trauma, frequency of exposure determines frequency range of sensitivity loss
- sudden, permanent SNHL caused by single exposure to an intense sound
- impulse sound, 130-140dB
- notch at 4kHz
tinnitus:
- auditory sensation in the absence of auditory input
- can be induced by ototoxic drugs
- thought that inhibition is decreased
- disinhibition
- GABA is inhibitory (glutamate is excitatory)
- perhaps reduction of GABA in certain structures may give rise to tinnitus
cochlear implant:
- microphone
- speech processor
- transmitter/receiver/stimulator
- electrodes
- pattern of stimulation mimics phase-locking
1.Hearing loss can arise in the conductive apparatus before the oval window, when it is known as conductive loss. Alternatively, it may arise in the cochlea or more centrally, when it is known as sensorineural hearing loss. If it arises in the cochlea, it is known as sensorineural hearing loss of cochlear origin.
2. Sensorineural hearing loss of cochlear origin can be caused by ototoxic drugs. Aminoglycoside antibiotics are ototoxic and have been extensively investigated; they damage hair cells, and particularly outer hair cells, in the high-frequency parts of the cochlea. They are likely to cause their damage at least in part by oxidative mechanisms, and antioxidants can provide some protection.
3. Other ototoxic agents include Cisplatin (an anticancer drug), some loop diuretics (bumetanide, furosemide and ethacrynic acid), aspirin (reversibly) and some organic solvents (e.g. styrene and toluene).
4. With acoustic overstimulation, for milder degrees of damage, there is minor disruption of the stereocilia. These degrees of damage are probably reversible. Greater degrees of damage cause loss of the hair cells.
5. Some forms of hearing losses have genetic causes. The most common inherited hearing loss, accounting for some 35% of all congenital (present from birth) hearing losses (depending on the population), is the type known as DFNB1. DFNB1 is caused by a mutation in the gene coding for the gap junction protein connexin-26. Patients with this mutation have problems cycling K+ from the cochlear scala through the cochlear walls back to the stria vascularis. This and many other inherited hearing losses have been invaluable for revealing basic mechanisms of the inner ear.
6. Mutations in the mitochondrial genome may also give rise to hearing loss. These syndromes are maternally inherited, commonly affect multiple organ systems and tend to increase with severity over time.
7. Aging causes sensorineural hearing loss (presbycusis), which can have both cochlear and central components. In the cochlea, vulnerable sites are the hair cells, the nerve and the stria vascularis.
8. Damage to the outer hair cells causes a loss of the active amplification of the traveling wave in the cochlear duct. This leads to a reduction in the magnitude of the traveling wave and to a broadening of the peak of the wave. The changes lead to a loss in the sensitivity of auditory nerve fibers, and a broadening of their tuning curves, that is to a loss of frequency resolution. Damage to the inner hair cells leads to a loss of sensitivity of the auditory nerve fibers without changes in frequency resolution.
9. The psychophysical results fit in with the physiological results. In sensorineural loss of cochlear origin, a loss of frequency resolution is often seen in addition to the loss in sensitivity. The loss in frequency resolution can be seen in widened psychophysical tuning curves and in widened psychophysical filters (critical bands). These factors lead to a particular difficulty in understanding broadband complex sounds, such as speech, and particularly against noisy backgrounds.
10. Some forms of tinnitus, that is those arising in the cochlea from the active mechanical amplification of the traveling wave, may give rise to objectively measurable sound emissions in the ear canal. However, this is unlikely to explain the clinically most important forms of tinnitus. Tinnitus is commonly associated with some degree of hearing loss. This suggests that is arises from a hypersensitivity of the central auditory nervous system when it is deprived of hypersensitivity of the central auditory nervous system when it is deprived of its normal input. It appears that neural activity increases in the auditory pathway, starting at the dorsal cochlear nucleus, and at later stages. The tinnitus may arise from a reduction in inhibition by the neurotransmitters GABA and glycine, and the anti-epileptic drugs vigabatrin and gabapentin that increase GABA inhibition have been reported to reverse tinnitus in experimental animals and some patients. Tinnitus may also arise from down regulation of K+ channels in the nucleus. where there is no effective drug treatment, the behavioral method known as "tinnitus retraining therapy" has proved effective in many patients.
11. With profound sensorineural hearing loss of cochlear origin, hearing can often be restored by a cochlear prosthesis. Current prosthesis consist of multiple electrodes inserted deep into the cochlear duct, where they can stimulate what cells remain of the auditory nerve. Stimuli are pre-processed to pick out the major spectral components of the stimuli, and electrodes at the appropriate positions in the cochlea (depending on the tonotopicity of the cochlea) are stimulated electrically. The amplitude of stimulation depends on the amplitude of the relevant spectral component at each moment, so that the final pattern of stimulation is able to mirror both the place-frequency and temporal aspects of cochlear function. After practice, good communication is achieved with running speech, and 80-90% of open-set sentences are understood correctly.
12. Attempts are being made to replace damaged cells in the inner ear, including the hair cells, cells of the spiral ganglion (auditory nerve) and the stria vascularis. These include stimulating the division of the supporting cells, to produce new hair cels, inoculating the cochlea with a virus engineered to contain a gene Math-1, which encourages the supporting cells to trans-differentiate (transform without undergoing cell division) into hair cells and the injection of stem cells. The transdifferentiation of supporting cells induced by Math-1 has been able to partially restore some hearing in adult animals even after complete loss of all hair cells.However, these methods require some remaining differentiated supporting cells in the organ of Corti, which is not usually the case with advanced sensorineural hearing loss. Stem cells may however form a possible treatment for repair of the auditory nerve.
13. Gene therapy may be possible, where the function of a damaged gene is replaced by an introduced normal copy of the gene. Protection of hair cells may also be possible by inhibiting apoptotic (the death of cells that occurs as a normal and controlled part of an organism's growth or development. Also called programmed cell death) use of antioxidant therapy and manipulations which increase the ear's own biochemical protective mechanisms, for example those arising in the phenomenon know as "sound conditioning" or "toughening", which work via corticosteroid defenses.
- Benign neuroma or schwannoma
- within cochlea
- acoustic trauma
- infections
- congenital anomalies
- age-related
- ototoxic agents; High Frequencies are lost first
- aminoglycosides
- antibiotics; gentamycin, kanamycin
- aspirin may counteract gentamycin
- taken up by hair cells
- cause damage via free radicals
- antibiotics; gentamycin, kanamycin
- loop diuretics
- salicylates
- aminoglycosides
- acoustic overstimulation
- genetic causes
cell death: Occurs by necrosis and apoptosis
Necrosis
- blebs form
- blebs fuse
- blebs rupture
- blebs form
- nucleus breaks apart
- cells breaks into bodies
mutations: 35% due to gene called connexin-26
- allow ions to pass between cells
- gap junctions in the stria vascularis
- causes severe, early onset hearing loss
presbycusis: SNHL due to aging
- sensory pattern associated with hair cell changes
- strial pattern associated with changes in the stria vascularis
- structural patterns with high energy consumption
- mitochondrial dysfunction
- HIGH FREQUENCIES ARE LOST BILATERALLY
noise induced oxidative cochlear injury
acoustic trauma: With Acoustic Trauma, frequency of exposure determines frequency range of sensitivity loss
- sudden, permanent SNHL caused by single exposure to an intense sound
- impulse sound, 130-140dB
- notch at 4kHz
tinnitus:
- auditory sensation in the absence of auditory input
- can be induced by ototoxic drugs
- thought that inhibition is decreased
- disinhibition
- GABA is inhibitory (glutamate is excitatory)
- perhaps reduction of GABA in certain structures may give rise to tinnitus
cochlear implant:
- microphone
- speech processor
- transmitter/receiver/stimulator
- electrodes
- pattern of stimulation mimics phase-locking